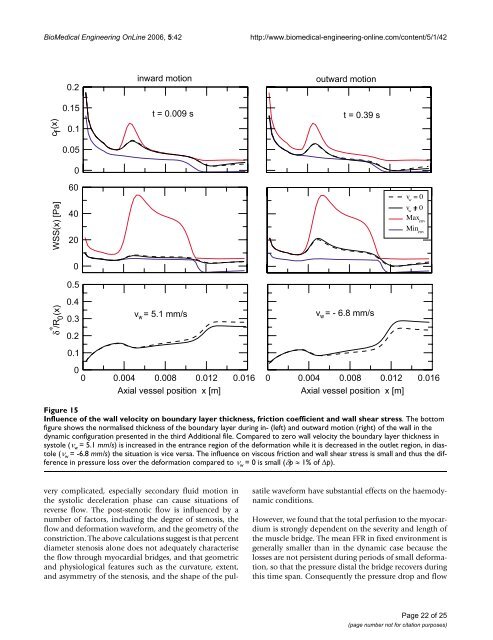
BioMedical Engineering OnLine 2006, 5:42http://www.biomedical-engineering-online.com/content/5/1/42 Figure Influence 15of <strong>the</strong> wall velocity on <strong>boundary</strong> <strong>layer</strong> thickness, friction coefficient and wall shear stressInfluence of <strong>the</strong> wall velocity on <strong>boundary</strong> <strong>layer</strong> thickness, friction coefficient and wall shear stress. The bot<strong>to</strong>mfigure shows <strong>the</strong> normalised thickness of <strong>the</strong> <strong>boundary</strong> <strong>layer</strong> during in- (left) and outward motion (right) of <strong>the</strong> wall in <strong>the</strong>dynamic configuration presented in <strong>the</strong> third Additional file. Compared <strong>to</strong> zero wall velocity <strong>the</strong> <strong>boundary</strong> <strong>layer</strong> thickness insys<strong>to</strong>le (ν w = 5.1 mm/s) is increased in <strong>the</strong> entrance region of <strong>the</strong> deformation while it is decreased in <strong>the</strong> outlet region, in dias<strong>to</strong>le(ν w = -6.8 mm/s) <strong>the</strong> situation is vice versa. The influence on viscous friction and wall shear stress is small and thus <strong>the</strong> differencein pressure loss over <strong>the</strong> deformation compared <strong>to</strong> ν w = 0 is small (δp ≈ 1% of Δp).very complicated, especially secondary fluid motion in<strong>the</strong> sys<strong>to</strong>lic deceleration phase can cause situations ofreverse flow. The post-stenotic flow is influenced by anumber of fac<strong>to</strong>rs, including <strong>the</strong> degree of stenosis, <strong>the</strong>flow and deformation waveform, and <strong>the</strong> geometry of <strong>the</strong>constriction. The above calculations suggest is that percentdiameter stenosis alone does not adequately characterise<strong>the</strong> flow through myocardial bridges, and that geometricand physiological features such as <strong>the</strong> curvature, extent,and asymmetry of <strong>the</strong> stenosis, and <strong>the</strong> shape of <strong>the</strong> pulsatilewaveform have substantial effects on <strong>the</strong> haemodynamicconditions.However, we found that <strong>the</strong> <strong>to</strong>tal perfusion <strong>to</strong> <strong>the</strong> myocardiumis strongly dependent on <strong>the</strong> severity and length of<strong>the</strong> muscle bridge. The mean FFR in fixed environment isgenerally smaller than in <strong>the</strong> dynamic case because <strong>the</strong>losses are not persistent during periods of small deformation,so that <strong>the</strong> pressure distal <strong>the</strong> bridge recovers duringthis time span. Consequently <strong>the</strong> pressure drop and flowPage 22 of 25(page number not for citation purposes)
BioMedical Engineering OnLine 2006, 5:42http://www.biomedical-engineering-online.com/content/5/1/42reduction across fixed stenoses are more pronounced thanin dynamic environments.As previously noted [8] found that <strong>the</strong> pressure proximal<strong>to</strong> <strong>the</strong> myocardial bridge was higher than <strong>the</strong> aortic pressure,and concluded that <strong>the</strong> disturbance of blood flowand high wall stress proximal <strong>to</strong> <strong>the</strong> myocardial bridgewas <strong>the</strong> main contribu<strong>to</strong>r <strong>to</strong> <strong>the</strong> development of a<strong>the</strong>rosclerosisin <strong>the</strong> proximal segment. The observed wall shearstress distributions indicate that <strong>the</strong> proximal segment ismore susceptible <strong>to</strong> <strong>the</strong> development of a<strong>the</strong>rosclerosis,firstly because <strong>the</strong> pressure is increased <strong>the</strong>re and secondlyfor reasons that <strong>the</strong> wall shear stress and <strong>the</strong>ir oscillationsare maximum in <strong>the</strong> entrance region of <strong>the</strong> deformation.In contrast bridged segments are relatively spared because<strong>the</strong> wall shear stress fades <strong>to</strong>wards <strong>the</strong> end of <strong>the</strong> deformation.In a series of myocardial bridges it is likely that <strong>the</strong>intima between <strong>the</strong> deformations and distal <strong>to</strong> <strong>the</strong> myocardialbridge are protected from a<strong>the</strong>rosclerosis because<strong>the</strong> wall shear stress is very low and negative in separatedflow regions.ConclusionWe have presented a <strong>method</strong> for simulation of unsteadyblood flow in a time dependent vessel geometry using an<strong>integral</strong> <strong>boundary</strong> <strong>layer</strong> <strong>method</strong>. The strong interaction of<strong>the</strong> viscous <strong>boundary</strong> <strong>layer</strong> and <strong>the</strong> inviscid core flow proposedby Veldman [58] models <strong>the</strong> pressure and <strong>the</strong>extent of <strong>the</strong> separation region by assuming Falkner-Skanflow profiles. The equations were modified <strong>to</strong> <strong>the</strong> flow situationunder consideration. Numerical simulations wereperformed for idealised stenosis geometries with a timedependent, smooth wall con<strong>to</strong>ur, but with a physiologicallyrealistic coronary artery flow waveform. The predictedvalues of fractional flow reserve in dynamic lesionsagree well with <strong>the</strong> clinical findings in [38], however, fur<strong>the</strong>rquantification in more defined geometries isrequired.Regarding <strong>the</strong> wall shear stresses and <strong>the</strong> development ofa<strong>the</strong>rosclerosis <strong>the</strong> findings are consistent with [1], where<strong>the</strong> intima beneath <strong>the</strong> bridge is protected from a<strong>the</strong>rosclerosis,and <strong>the</strong> proximal segment is more susceptible <strong>to</strong><strong>the</strong> development of a<strong>the</strong>rosclerotic lesions.Besides <strong>the</strong> advantage of computational time taken for <strong>the</strong>simulation, <strong>the</strong> choice of parameters, such as location,length and severity of <strong>the</strong> lesion are easily determined bycoronary angiography. Due <strong>to</strong> <strong>the</strong> assumptions made in<strong>the</strong> <strong>boundary</strong> <strong>layer</strong> model, <strong>the</strong> approximation fails for <strong>the</strong>prediction of reverse flow and flow where <strong>the</strong> <strong>boundary</strong><strong>layer</strong>s merge, i.e. fully developed flow. Under <strong>the</strong>seaspects severe deformations cannot be <strong>calculate</strong>d, because<strong>the</strong> <strong>boundary</strong> <strong>layer</strong>s merge in <strong>the</strong> deformation. Fur<strong>the</strong>r,<strong>the</strong> length of <strong>the</strong> computational domain is restricted by<strong>the</strong> entrance length, which depends on Reynolds number.And finally <strong>the</strong> pulsatile frequency and <strong>the</strong> frequency ofwall motion has <strong>to</strong> be sufficiently low (a few Hz), so that<strong>the</strong> approximation of quasi-stationary evolution of <strong>the</strong><strong>boundary</strong> <strong>layer</strong>s is satisfied.We believe that <strong>the</strong> parameters and equations in this articleare detailed enough <strong>to</strong> describe <strong>the</strong> physiological consequencesalso in a clinical setting, however, this remains<strong>to</strong> be confirmed by in vivo studies. The functional consequence,especially for severe sys<strong>to</strong>lic compression, is consistentwith clinical findings published in <strong>the</strong> literature [1-3,6,8,38,51], where myocardial bridging is found <strong>to</strong> beresponsible for myocardial ischaemia. The comparison ofour findings with <strong>the</strong> published data from patient studiesin [38,51] supports a potential clinical relevance of oursimulation.Authors' contributionsSB developed <strong>the</strong> <strong>boundary</strong> <strong>layer</strong> model, carried out <strong>the</strong>simulations and drafted <strong>the</strong> manuscript. SM drafted <strong>the</strong>section on <strong>the</strong> clinical situation. AT participated in modeldevelopment in its design and coordination. All authorsread and approved <strong>the</strong> final manuscript.Additional materialAdditional file 1Animation of <strong>the</strong> left descending coronary artery assuming Hagen-Poiseuille flow. The animation shows <strong>the</strong> pressure in <strong>the</strong> main segmentsof <strong>the</strong> left coronary arterial tree during one pulsatile cycle. The unit of <strong>the</strong>colour bar is kPa, <strong>the</strong> playback speed is slower by a fac<strong>to</strong>r of three thanrealtime.Click here for file[http://www.biomedcentral.com/content/supplementary/1475-925X-5-42-S1.mov]Additional file 2Animation of a double myocardial bridge in <strong>the</strong> left descending coronaryartery assuming Hagen-Poiseuille flow. As in <strong>the</strong> first animation<strong>the</strong> pressure in <strong>the</strong> main segments of <strong>the</strong> left coronary arterial tree areshown over one heart cycle. A series of two myocardial bridges with length8 mm and deformation ζ 0 = 0.25 are located in <strong>the</strong> mid LAD. The pressuredrop is underestimated by <strong>the</strong> assumption of Hagen-Poiseuille flow,consequently perfusion <strong>to</strong> <strong>the</strong> myocardium remains nearly unaffected. Theunit of <strong>the</strong> colour bar is kPa, <strong>the</strong> playback speed is slower by a fac<strong>to</strong>r ofthree than realtime.Click here for file[http://www.biomedcentral.com/content/supplementary/1475-925X-5-42-S2.mov]Page 23 of 25(page number not for citation purposes)
- Page 1 and 2: BioMedical Engineering OnLineBioMed
- Page 3: BioMedical Engineering OnLine 2006,
- Page 6 and 7: BioMedical Engineering OnLine 2006,
- Page 8 and 9: BioMedical Engineering OnLine 2006,
- Page 10 and 11: BioMedical Engineering OnLine 2006,
- Page 12: BioMedical Engineering OnLine 2006,
- Page 15 and 16: BioMedical Engineering OnLine 2006,
- Page 17 and 18: BioMedical Engineering OnLine 2006,
- Page 19 and 20: BioMedical Engineering OnLine 2006,
- Page 21: BioMedical Engineering OnLine 2006,
- Page 25: BioMedical Engineering OnLine 2006,