4410 J. Med. Plants Res. ovarian cancer cell lines (SKOV3) and human cervical cancer cell lines (Hela) in vitro. B. yinchowense is abundantly distributed in the Northwest of China and widely used in Chinese folk medicine. No evidence is available in the literatures concerning its constituents and pharmacological activities. To systematically evaluate its potential anticancer activity, the constituents were studied by activity-guided fraction and result in the isolation of 13 saikosaponins. Their structures were identified on the basis of spectral data. The isolation, structure elucidation and evaluation for cytotoxic activities were described in this study. MATERIALS AND METHODS General experimental procedure Nuclear magnetic resonance (NMR) spectra were recorded on a Brucker DRX-500 spectrometer (Brucker Biosciences Corporation, Billerica, MA) with tetramethylsilane (TMS) as internal standard operating at 500 and 125 MHz for 1 H and 13 C, respectively. Fast atom bombardment-mass spectra (FAB-MS) and high resolutionfast atom bombardment-mass spectra (HR-FABMS) were recorded on a Micromass Autospec-Q instrument (Micromass Ltd., Manchester, UK). Infrared (IR) spectra were recorded in KBr discs using a Perkin-Elmer 983G spectrophotometer (Perkin-Elmer Ltd., USA). Gas chromatography (GC) analysis was carried out on an Agilent 6890N gas chromatography (Agilent Co., USA) using an HP-5 capillary column. Column chromatography was performed with silica gel (100 to 200 mesh, Qingdao Marine Chemical Co., Qingdao, P. R. China), Sephadex LH-20 (25 to 100 μm, GE Healthcare Bioscences AB, Uppsaki, Sweden), octadecyl silica (25 to 40 μm, Merck, USA), D101 macroporous resins (Tianjin Gujiao Factory, Tianjin, P. R. China), MCI Gel CHP20P (75 to 150 µm, Mitsubishi Chemical, Japan). Thin layer chromatography (TLC) was performed on precoated silica gel GF254 (0.2 mm thick, Qingdao Marine Chemical Co., Qingdao, P. R. China) and spots were detected by spraying with 10% ethanolic H2SO4 reagent. 3-(4, 5dimethylthiiazol-2yl)-2,5-diphenyl tetrazolium bromide (MTT) was purchased from Sigma-Aldrich (St. Louis, MO, USA). Plant collection The roots of B. yinchowense were collected from Dingxi County, Gansu province, China, in August, 2009 and identified by the author, Professor Ruile Pan of the Institute of Medicinal Plant, Chinese Academy of Medical Sciences and Peking Union Medical College, Beijing, China, where a voucher specimen (No. 20090815) was deposited. Extraction The dried and powdered roots (800 g) of B. yinchowense were extracted with 60% ethanol containing 0.5% ammoniae aqua (three times, 1 L each) at room temperature for 12 h. The ethanol extracts were combined and evaporated in vacuo, to yield a dark brown residue (120 g), which was dissolved in H2O-MeOH (5:95) solution (200 ml), and then portioned with n-hexane of 200 ml) to get the nhexane-soluble fraction. The H2O-MeOH (5:95) layer was evaporated to remove residual MeOH, and then distilled water (200 ml) was added. This aqueous solution was subjected to a column contained 1 kg D101 macroporous resin and was eluted successively with water (2 L), 90% ethanol (2 L), respectively. Evaporation of the respective solvents gave n-hexane (12 g), water (42 g) and 90% ethanol (32 g) fractions. The 90% ethanol fraction is saponin-enriched part. Each fraction was evaluated for the cytotoxic activity on the tumor cell lines (Table 1). It was shown that the activity resided in the saponin-enriched part. Isolation Saponin-enriched part (32 g) was subjected to MCI column, eluting with a gradient of water-methanol (from 100:0 to 5:95), to yield 5 fractions. Fraction 2 (8 g) was chromatographed repeatedly on silica gel using chloroform-methanol (8:2) and octadecylsilane (ODS)-C18 with the elution of methanol-water (7:3) to afford 1 (80 mg),2 (55 mg),3 (75 mg), 11 (5 mg) and 9 (17 mg). Fraction 3 (10 g) was separated into three sub-fractions by ODS column using methanol-water (7:3) as elution and the second sub-fraction was subjected to repeated column chromatography, first on silica gel, chloroform:methanol (8:2) and purified on pharmadex LH-20 (methanol) to obtain 4 (51 mg), 5 (38 mg), 6 (20 mg) and 12 (8 mg). Compound 7 (35 mg), 8 (37 mg), 13 (6 mg) and compound 10 (22 mg) were purified from Fraction 4 (5 g) by repeated semipreparative high performance liquid chromatography (HPLC) using methanolwater (60:40) as fluent. Characterization of isolation Saikosaponin a (1): White amorphous powder, mp 229 to 230°C, 1 H-NMR (C5D5N, 500 MHz): � 0.91, 0.92, 0.99, 1.12, 1.31, 1.37 (each 3H, s, tert-Me×6), 1.47 (3H, d, J=6.6 Hz, Fuc-CH3), 4.94 (1H, d, J=7.8 Hz, Fuc-1′-H), 5.35 (1H, d, J=8.4 Hz, Glc-1″-H), 5.66 (1H, dd, J=10.2, 2.4 Hz, 11-H), 6.00 (1H, d, J=10.2 Hz, 12-H). 13 C-NMR data (Table 1). Saikosaponin c (2): White amorphous powder, mp 207 to 209°C, 1 H-NMR (C5D5N, 500 MHz): � 0.86, 0.91, 0.96, 0.97, 1.14, 1.28, 1.35 (each 3H, s, tert-Me × 7), 1.66 (3H, d, J=6.0 Hz, Rha-CH3), 4.94 (1H, d, J=7.8 Hz, Glc-1′-H), 4.52 (1H, d, J=9.0 Hz, Rha-1″-H), 4.79 (1H, d, J =7.8 Hz, Glc-1′″-H), 5.64 (1H, dd, J=10.2, 2.4 Hz, 11- H), 5.90 (1H, d, J=10.2 Hz, 12-H). 13 C-NMR data (Table 1). Saikosaponin d (3): White amorphous powder, mp 227 to 228°C, 1 H-NMR (C5D5N, 500 MHz): � 0.91, 0.92, 0.99, 1.12, 1.31, 1.37 (each 3H, s, tert-Me×6), 1.47 (3H, d, J=6.6 Hz, Fuc-CH3), 4.94 (1H, d, J=7.8 Hz, Fuc-1′-H), 5.35 (1H, d, J=8.4 Hz, Glc-1″-H), 5.66 (1H, dd, J=10.2, 3.0 Hz, 11-H), 6.02 (1H, d, J=10.2 Hz, 12-H). 13 C-NMR data (Table 1). Saikosaponin b2 (4): White amorphous powder, mp 201 to 203°C, 1 H-NMR (C5D5N, 500 MHz): � 0.89, 0.92, 0.99, 1.01, 1.04, 1.68 (each 3H, s, tert-Me×6), 1.45 (3H, d, J=6.0 Hz, Fuc-CH3), 4.99 (1H, d, J=7.8 Hz, Fuc-1′-H), 5.40 (1H, d, J=7.8 Hz, Glc-1″-H), 6.70 (1H, dd, J=10.2, 1.8 Hz, 11-H), 5.72 (1H, d, J=10.2 Hz, 12-H). 13 C-NMR data (Table 1). Saikosaponin f (5): White amorphous powder, mp198 to 200°C 1 H-NMR (C5D5N, 500 MHz): � 0.82, 0.95, 0.99, 1.00, 1.01, 1.29, 1.35 (each 3H, s, tert-Me×7), 1.65 (3H, d, J=6.0 Hz, Rha-CH3), 4.94 (1H, d, J=7.8 Hz, Glc-1′-H), 4.52 (1H, d, J=9.0 Hz, Rha-1″-H), 4.79 (1H, d, J=7.8 Hz, Glc-1′″-H), 5.86 (1H, br, 12-H). 13 C-NMR data (Table 1). Saikosaponin b4 (6): White amorphous powder, mp 206 to 208°C, 1 H-NMR (C5D5N, 500 MHz): � 0.97, 1.01, 1.01, 1.12, 1.14, 1.88 (each 3H, s, tert-Me×6), 3.26 (3H, s, OCH3), 1.45 (3H, d, J=6.0 Hz, Fuc-CH3), 4.96 (1H, d, J=7.2 Hz, Fuc-1′-H), 5.33 (1H, d, J=7.8 Hz, Glc-1″-H), 5.60 (1H, d, J=3.0 Hz, 12-H). 13 C-NMR data (Table 1). 6″-O-acetylsaikosaponin a (7): White amorphous powder, mp 204 to 205°C, 1 H-NMR (C5D5N, 500 MHz): � 0.89, 0.92, 0.92, 0.98,
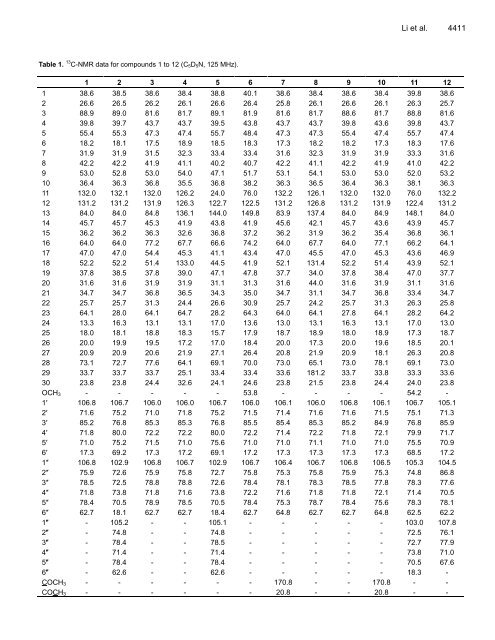
Table 1. 13 C-NMR data for compounds 1 to 12 (C5D5N, 125 MHz). Li et al. 4411 1 2 3 4 5 6 7 8 9 10 11 12 1 38.6 38.5 38.6 38.4 38.8 40.1 38.6 38.4 38.6 38.4 39.8 38.6 2 26.6 26.5 26.2 26.1 26.6 26.4 25.8 26.1 26.6 26.1 26.3 25.7 3 88.9 89.0 81.6 81.7 89.1 81.9 81.6 81.7 88.6 81.7 88.8 81.6 4 39.8 39.7 43.7 43.7 39.5 43.8 43.7 43.7 39.8 43.6 39.8 43.7 5 55.4 55.3 47.3 47.4 55.7 48.4 47.3 47.3 55.4 47.4 55.7 47.4 6 18.2 18.1 17.5 18.9 18.5 18.3 17.3 18.2 18.2 17.3 18.3 17.6 7 31.9 31.9 31.5 32.3 33.4 33.4 31.6 32.3 31.9 31.9 33.3 31.6 8 42.2 42.2 41.9 41.1 40.2 40.7 42.2 41.1 42.2 41.9 41.0 42.2 9 53.0 52.8 53.0 54.0 47.1 51.7 53.1 54.1 53.0 53.0 52.0 53.2 10 36.4 36.3 36.8 35.5 36.8 38.2 36.3 36.5 36.4 36.3 38.1 36.3 11 132.0 132.1 132.0 126.2 24.0 76.0 132.2 126.1 132.0 132.0 76.0 132.2 12 131.2 131.2 131.9 126.3 122.7 122.5 131.2 126.8 131.2 131.9 122.4 131.2 13 84.0 84.0 84.8 136.1 144.0 149.8 83.9 137.4 84.0 84.9 148.1 84.0 14 45.7 45.7 45.3 41.9 43.8 41.9 45.6 42.1 45.7 43.6 43.9 45.7 15 36.2 36.2 36.3 32.6 36.8 37.2 36.2 31.9 36.2 35.4 36.8 36.1 16 64.0 64.0 77.2 67.7 66.6 74.2 64.0 67.7 64.0 77.1 66.2 64.1 17 47.0 47.0 54.4 45.3 41.1 43.4 47.0 45.5 47.0 45.3 43.6 46.9 18 52.2 52.2 51.4 133.0 44.5 41.9 52.1 131.4 52.2 51.4 43.9 52.1 19 37.8 38.5 37.8 39.0 47.1 47.8 37.7 34.0 37.8 38.4 47.0 37.7 20 31.6 31.6 31.9 31.9 31.1 31.3 31.6 44.0 31.6 31.9 31.1 31.6 21 34.7 34.7 36.8 36.5 34.3 35.0 34.7 31.1 34.7 36.8 33.4 34.7 22 25.7 25.7 31.3 24.4 26.6 30.9 25.7 24.2 25.7 31.3 26.3 25.8 23 64.1 28.0 64.1 64.7 28.2 64.3 64.0 64.1 27.8 64.1 28.2 64.2 24 13.3 16.3 13.1 13.1 17.0 13.6 13.0 13.1 16.3 13.1 17.0 13.0 25 18.0 18.1 18.8 18.3 15.7 17.9 18.7 18.9 18.0 18.9 17.3 18.7 26 20.0 19.9 19.5 17.2 17.0 18.4 20.0 17.3 20.0 19.6 18.5 20.1 27 20.9 20.9 20.6 21.9 27.1 26.4 20.8 21.9 20.9 18.1 26.3 20.8 28 73.1 72.7 77.6 64.1 69.1 70.0 73.0 65.1 73.0 78.1 69.1 73.0 29 33.7 33.7 33.7 25.1 33.4 33.4 33.6 181.2 33.7 33.8 33.3 33.6 30 23.8 23.8 24.4 32.6 24.1 24.6 23.8 21.5 23.8 24.4 24.0 23.8 OCH3 - - - - - 53.8 - - - - 54.2 - 1′ 106.8 106.7 106.0 106.0 106.7 106.0 106.1 106.0 106.8 106.1 106.7 105.1 2′ 71.6 75.2 71.0 71.8 75.2 71.5 71.4 71.6 71.6 71.5 75.1 71.3 3′ 85.2 76.8 85.3 85.3 76.8 85.5 85.4 85.3 85.2 84.9 76.8 85.9 4′ 71.8 80.0 72.2 72.2 80.0 72.2 71.4 72.2 71.8 72.1 79.9 71.7 5′ 71.0 75.2 71.5 71.0 75.6 71.0 71.0 71.1 71.0 71.0 75.5 70.9 6′ 17.3 69.2 17.3 17.2 69.1 17.2 17.3 17.3 17.3 17.3 68.5 17.2 1″ 106.8 102.9 106.8 106.7 102.9 106.7 106.4 106.7 106.8 106.5 105.3 104.5 2″ 75.9 72.6 75.9 75.8 72.7 75.8 75.3 75.8 75.9 75.3 74.8 86.8 3″ 78.5 72.5 78.8 78.8 72.6 78.4 78.1 78.3 78.5 77.8 78.3 77.6 4″ 71.8 73.8 71.8 71.6 73.8 72.2 71.6 71.8 71.8 72.1 71.4 70.5 5″ 78.4 70.5 78.9 78.5 70.5 78.4 75.3 78.7 78.4 75.6 78.3 78.1 6″ 62.7 18.1 62.7 62.7 18.4 62.7 64.8 62.7 62.7 64.8 62.5 62.2 1″′ - 105.2 - - 105.1 - - - - - 103.0 107.8 2″′ - 74.8 - - 74.8 - - - - - 72.5 76.1 3″′ - 78.4 - - 78.5 - - - - - 72.7 77.9 4″′ - 71.4 - - 71.4 - - - - - 73.8 71.0 5″′ - 78.4 - - 78.4 - - - - - 70.5 67.6 6″′ - 62.6 - - 62.6 - - - - - 18.3 - COCH3 - - - - - - 170.8 - - 170.8 - - COCH3 - - - - - - 20.8 - - 20.8 - -