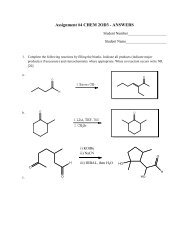
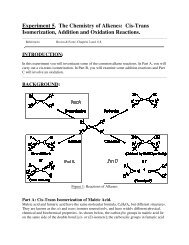
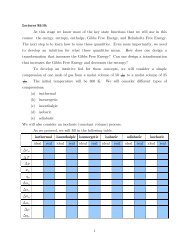
Organic Chemistry Structures of Organic Compounds
Organic Chemistry Structures of Organic Compounds
Organic Chemistry Structures of Organic Compounds
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
Less stableMore stableCH 3 MgBr + H-OH → CH 3 -H + BrMgOHOther C-C Bond Forming Reactionsδ-δ+MgBr+RRδ+ δ-OetherRROMgBrH 3O +RROHketonealcoholNote: CAN’T DO THE FOLLOWING REACTION.δ-δ+MgBr+RRBretherRRHalkyl halideEthers are less polar than the alcohols (No OH’s for H-bonding)They are made in a similar fashion to alcoholsPreparation <strong>of</strong> ethers (5 S N 2)CHBr + 3O- Na + O + NaBrCH 314 Making Alkoxides (Reducing Metals)2OH + 2Na 2ONa + H 2Alkoxides are strong bases (stronger than hydroxide) and also good Lewis bases or nucleophilesRecall: 2HO-H + 2K → 2HO-K + H 2 ↑