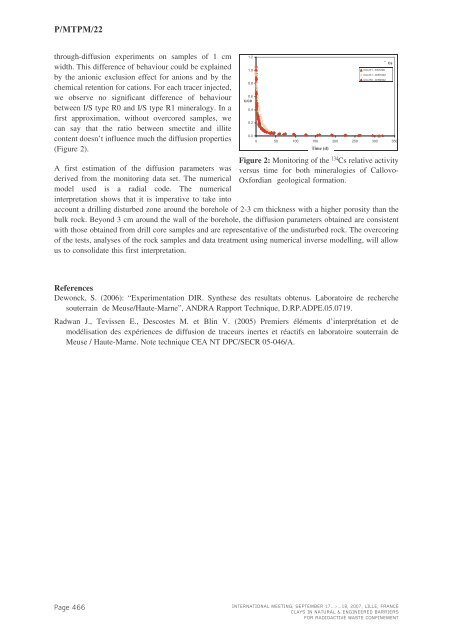
134P/<strong>MTPM</strong>/22through-diffusion experiments on samples of 1 cmwidth. This difference of behaviour could be explainedby the anionic exclusion effect for anions and by thechemical retention for cations. For each tracer injected,we observe no significant difference of behaviourbetween I/S type R0 and I/S type R1 mineralogy. In afirst approximation, without overcored samples, wecan say that the ratio between smectite and illitecontent doesn’t influence much the diffusion properties(Figure 2).A first estimation of the diffusion parameters wasderived from the monitoring data set. The numericalmodel used is a radial code. The numericalinterpretation shows that it is imperative to take intoFigure 2: Monitoring of the 134 Cs relative activityversus time for both mineralogies of Callovo-Oxfordian geological formation.account a drilling disturbed zone around the borehole of 2-3 cm thickness with a higher porosity than thebulk rock. Beyond 3 cm around the wall of the borehole, the diffusion parameters obtained are consistentwith those obtained from drill core samples and are representative of the undisturbed rock. The overcoringof the tests, analyses of the rock samples and data treatment using numerical inverse modelling, will allowus to consolidate this first interpretation.1.21.00.80.6C/C00.40.20.0Cs0 50 100 150 200 250 300 350Time Temps (d)(j)COx R1- - EST208COx R1- - DIR1002COx R0- - DIR2002ReferencesDewonck, S. (2006): “Experimentation DIR. Synthese des resultats obtenus. Laboratoire de recherchesouterrain de Meuse/Haute-Marne”, ANDRA Rapport Technique, D.RP.ADPE.05.0719.Radwan J., Tevissen E., Descostes M. et Blin V. (2005) Premiers éléments d’interprétation et demodélisation des expériences de diffusion de traceurs inertes et réactifs en laboratoire souterrain deMeuse / Haute-Marne. Note technique CEA NT DPC/SECR 05-046/A.Page 466INTERNATIONAL MEETING, SEPTEMBER 17...>...18, 2007, LILLE, FRANCECLAYS IN NATURAL & ENGINEERED BARRIERSFOR RADIOACTIVE WASTE CONFINEMENT
P/<strong>MTPM</strong>/23INFLUENCE OF HUMIC COLLOIDSON THE MIGRATION OF U(VI)IN COMPACTED CLAYJ. Mibus 1, 2 , S. Sachs 11. Forschungszentrum Dresden-Rossendorf, Institute of Radiochemistry, P.O. Box 510119,D-01314 Dresden, Germany2. Paul Scherrer Institut, Laboratory for Waste Management, CH-5232 Villigen PSI, SwitzerlandClay minerals and organic substances are widespread components of many soils, sediments, and rocks. Ashumic acids (HA) possess a strong ability for metal complexation, they can influence the migration ofradioactive and toxic metal ions in clayey environments. However, the migration behavior of humiccolloids and its impact on the actinide migration in clay formations is poorly understood.Former studies discuss the influence of HA on the radionuclide migration in clays controversially. Woldand Eriksen [1] reported the diffusion of humic colloids through compacted bentonite without strongphysical hindrance mobilizing metal ions. In contrast, Wang et al. [2] found a significant immobilizationof Eu(III) by humic substances. Maes et al. [3] stressed the reversibility of the interaction betweenradionuclide, organic matter, and clay. The competition of the adsorbents for radionuclides is kinetic anddetermines their distribution between mobile and immobile phase.In the present study we investigated the diffusion behavior of humic colloids in compacted clay as afunction of compaction and pH. The impact on the uranium transport was studied by comparing theuranium diffusion in absence and presence of HA.Kaolinite KGa-1b and a 14 C-labeled synthetic HA type M42 [4] in a 0.01 M NaClO 4 / 1 mM NaN 3 mediumwere used. We studied steady-state through-diffusion applying a constant concentration gradient. The claywas compacted in diffusion cells to bulk dry densities ñ of 1.32 to 1.67 g/cm 3 and equilibrated at pH 5 or7. HA was used at a concentration of 12 mg/L. For the experiments involving U(VI), a UO 2+ 2 concentrationof 1·10 -6 M ( 238 U) was used, whereas 234 U was employed as tracer in the experiments with humicsubstances. The tracer activities in the high and low concentration reservoirs were periodically analyzedby liquid scintillation counting and by ICP-MS. After the end of the experiments, in-diffusion profiles ofHA and uranium were taken. Modeling based on Fick’s second law yields the effective diffusioncoefficient D e [m 2 /s] and the rock capacity factor α [-].The migration of HA in compacted kaolinite was found to be governed by diffusion. A constant diffusiveflux established after a transient state of 8 to 30 days. The size distribution of HA in the high and lowconcentration reservoir suggests that the mean particle size shifts from 50 kD in the high to less than 1 kDin the low concentration reservoir. Only small HA molecules are able to pass the kaolinite pore system,which demonstrates a filtration of colloidal particles in the clay.The effective diffusion coefficients decrease with increasing clay density. The values of HA (5.1 · 10 -12 m 2 /sto 1.2 · 10 -10 m 2 /s) are always lower than those of HTO ((1.6 - 2.7) · 10 -10 m 2 /s). The effective diffusioncoefficients of HA were found to be higher by a factor of four to six at pH 7 compared to pH 5. Here,differences in the free diffusion coefficients D 0 of the macro molecules are assumed. Lead et al. [5]observed an increase in the D 0 of Suwannee river humic acid by a factor of about 1.2 when increasing thepH from 5 to 7. This effect has been attributed to an aggregation of humic colloids at pH below 7.Surprisingly, a stronger adsorption of HA has been found at pH 7, where the electrostatic repulsionbetween the deprotonated HA and the negatively charged clay surface increases.INTERNATIONAL MEETING, SEPTEMBER 17...>...18, 2007, LILLE, FRANCECLAYS IN NATURAL & ENGINEERED BARRIERSFOR RADIOACTIVE WASTE CONFINEMENTPage 467