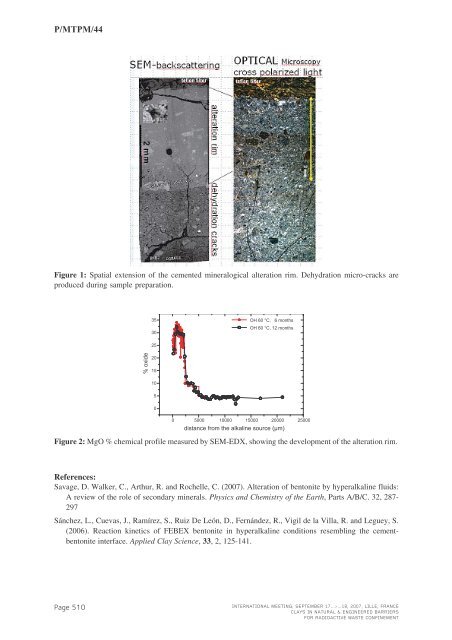
P/<strong>MTPM</strong>/44Figure 1: Spatial extension of the cemented mineralogical alteration rim. Dehydration micro-cracks areproduced during sample preparation.35 OH 60 °C, 6 months30OH 60 °C, 12 months25% oxide201510500 5000 10000 15000 20000 25000distance from the alkaline source (µm)Figure 2: MgO % chemical profile measured by SEM-EDX, showing the development of the alteration rim.References:Savage, D. Walker, C., Arthur, R. and Rochelle, C. (2007). Alteration of bentonite by hyperalkaline fluids:A review of the role of secondary minerals. Physics and Chemistry of the Earth , Parts A/B/C. 32, 287-297Sánchez, L., Cuevas, J., Ramírez, S., Ruiz De León, D., Fernández, R., Vigil de la Villa, R. and Leguey, S.(2006). Reaction kinetics of FEBEX bentonite in hyperalkaline conditions resembling the cementbentoniteinterface. Applied Clay Science , 33, 2, 125-141.Page 510INTERNATIONAL MEETING, SEPTEMBER 17...>...18, 2007, LILLE, FRANCECLAYS IN NATURAL & ENGINEERED BARRIERSFOR RADIOACTIVE WASTE CONFINEMENT
P/<strong>MTPM</strong>/45TRANSPORT NUMBER OF SODIUM IONSIN WATER-SATURATED, COMPACTEDNA-MONTMORILLONITETomohiro Higashihara *, Kumiko Kinoshita, Yorimasa Akagi, Seichi Sato, Tamotsu KozakiDivision of Energy and Environmental Systems, Graduate School of Engineering, HokkaidoUniversity, Kita 13 Nishi 8, Sapporo 060-8628, Japan* Present address, Civil Engineering Research Laboratory, Central Research Institute of ElectricPower Industry, 1646 Abiko, Abiko-shi, Chibaken 270-1194, JapanINTRODUCTIONIn geologic disposal of both high-level radioactive wastes and a part of TRU wastes, compacted bentoniteis a promising material to be used for a buffer material. Radionuclides are not migrated by advection butby diffusion, because of very low hydraulic conductivity of the bentonite. It is important to clarify thediffusion process of the radionuclide as a part of a safety assessment of the geologic disposal.In the buffer material, the diffusion of the radionuclides may be affected by other ions in the groundwater,because the nuclides as ions diffuse in electrically neutral condition by coupling with counter ions.However, there are quite few reports in which the contribution of these counter ions to diffusion of ionsis examined. Nakazawa et al. (1999) suggested that the charge carrier of electrical conduction in the Namontmorilloniteis hydrogen ions rather than Na + ions. However, the major charge carrier in the compactedbentonite has not been clarified sufficiently, because their conclusion was based on the indirect methodsuch as measurement of electrical conductivities. Measurement of transport number is one of directmethods and is one of the most basic electrochemical properties of materials. The transport numbersuggests the contribution of counter ions to the migration of radionuclides. The purpose of this study is topropose an experimental method to determine the transport number of the Na + ions in the water-saturated,compacted Na-montmorillonite to examine possible counter ions to the diffusion of radionuclides in themontmorillonite.EXPERIMENTALThe Na-montmorillonite powder was compacted in an acrylic resin cells, 20 mm in diameter and20 mm in height. The clay columns were saturated with 0.1 M NaClO 4 solution. The transport numberwas determined using a moving boundary method. To estimate the transport number of Na + ions inthe montmorillonite, two processes are concerned with. One is electromigration of Na + ions. Themigration of the Na + ions was traced, using 22 Na + spiked as a thin layer. The other is the electroosmoticflow of pore water, because it is possible that the Na + ions migrate together with pore water.The flow of pore water was traced using dissolved helium, because the helium can be regarded as anon-sorbing tracer. For measuring the electro-osmotic flow, a part of the prepared columns weresaturated with dissolved helium by bubbling. The osmotic flow was measured using an inflection pointof helium distribution in the montmorillonite (Higashihara et al., 2004).RESULTSDispersivity parameters, which were obtained in these experiments, were 10 -5 m for Na + and 10 -3 m for He.This indicates that a property of the migration path for Na + ions is deferent from one for He. It is quitepossible that, in the montmorillonite, the Na + ions mainly migrate in interlayer space and/or on outersurface, and the He does in pore space, as was suggested previously by Higashihara et al. (2004).Therefore, it is considered that the migration of the Na + is not affected by the electro-osmotic flow. Thetransport numbers of Na + ions were determined taking no account of the contribution of the electro-osmoticflow.INTERNATIONAL MEETING, SEPTEMBER 17...>...18, 2007, LILLE, FRANCECLAYS IN NATURAL & ENGINEERED BARRIERSFOR RADIOACTIVE WASTE CONFINEMENTPage 511