Ch18 Amines(pdf) - KFUPM Open Courseware
Ch18 Amines(pdf) - KFUPM Open Courseware
Ch18 Amines(pdf) - KFUPM Open Courseware
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
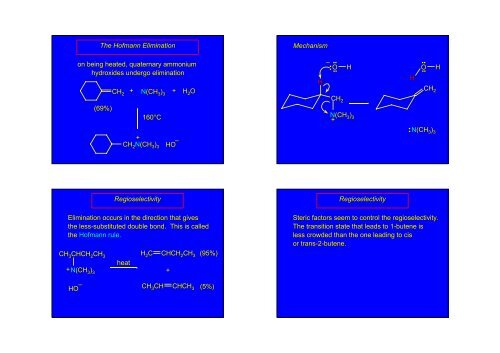
The Hofmann EliminationMechanismon being heated, quaternary ammoniumhydroxides undergo eliminationCH 2+ N(CH3 ) 3+ H 2 OH–••••O••HHO ••••CH 2H(69%)160°CCH 2N(CH+ 3 ) 3+CH 2 N(CH3 ) 3HO –•• N(CH3 ) 3RegioselectivityRegioselectivityElimination occurs in the direction that givesthe less-substituted substituted double bond. This is calledthe Hofmann rule.CH 3 CHCH 2 CH+ N(CH3 ) 3CH 3H 2 C CHCH 2 CH 3heat+(95%)Steric factors seem to control the regioselectivity.The transition state that leads to 1-butene 1isless crowded than the one leading to cisor trans-2-butene.HO –CH 3 CHCHCH 3(5%)