Photochemistry and Photophysics of Coordination Compounds
Photochemistry and Photophysics of Coordination Compounds
Photochemistry and Photophysics of Coordination Compounds
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
96 N. Armaroli et al.<br />
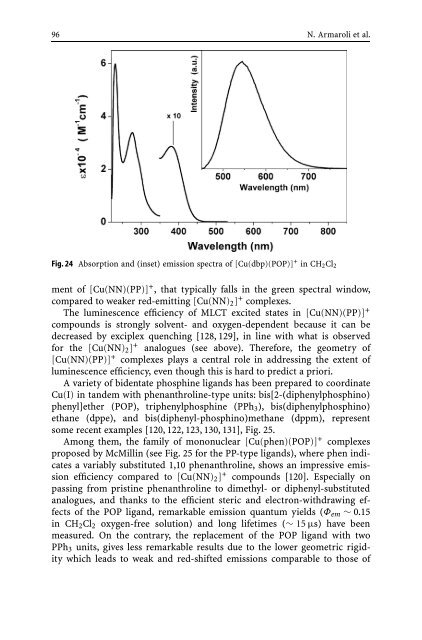
Fig. 24 Absorption <strong>and</strong> (inset) emission spectra <strong>of</strong> [Cu(dbp)(POP)] + in CH2Cl2<br />
ment <strong>of</strong> [Cu(NN)(PP)] + , that typically falls in the green spectral window,<br />
compared to weaker red-emitting [Cu(NN)2] + complexes.<br />
The luminescence efficiency <strong>of</strong> MLCT excited states in [Cu(NN)(PP)] +<br />
compounds is strongly solvent- <strong>and</strong> oxygen-dependent because it can be<br />
decreased by exciplex quenching [128, 129], in line with what is observed<br />
for the [Cu(NN)2] + analogues (see above). Therefore, the geometry <strong>of</strong><br />
[Cu(NN)(PP)] + complexes plays a central role in addressing the extent <strong>of</strong><br />
luminescence efficiency, even though this is hard to predict a priori.<br />
A variety <strong>of</strong> bidentate phosphine lig<strong>and</strong>s has been prepared to coordinate<br />
Cu(I) in t<strong>and</strong>em with phenanthroline-type units: bis[2-(diphenylphosphino)<br />
phenyl]ether (POP), triphenylphosphine (PPh3), bis(diphenylphosphino)<br />
ethane (dppe), <strong>and</strong> bis(diphenyl-phosphino)methane (dppm), represent<br />
some recent examples [120, 122, 123, 130, 131], Fig. 25.<br />
Among them, the family <strong>of</strong> mononuclear [Cu(phen)(POP)] + complexes<br />
proposed by McMillin (see Fig. 25 for the PP-type lig<strong>and</strong>s), where phen indicates<br />
a variably substituted 1,10 phenanthroline, shows an impressive emission<br />
efficiency compared to [Cu(NN)2] + compounds [120]. Especially on<br />
passing from pristine phenanthroline to dimethyl- or diphenyl-substituted<br />
analogues, <strong>and</strong> thanks to the efficient steric <strong>and</strong> electron-withdrawing effects<br />
<strong>of</strong> the POP lig<strong>and</strong>, remarkable emission quantum yields (Φem ∼ 0.15<br />
in CH2Cl2 oxygen-free solution) <strong>and</strong> long lifetimes (∼ 15 µs) have been<br />
measured. On the contrary, the replacement <strong>of</strong> the POP lig<strong>and</strong> with two<br />
PPh3 units, gives less remarkable results due to the lower geometric rigidity<br />
which leads to weak <strong>and</strong> red-shifted emissions comparable to those <strong>of</strong>