- Page 1 and 2: SYNTHESIS AND IN VITRO PHARMACOLOGY
- Page 3 and 4: Promotor : prof.dr H. Timmerman Cop
- Page 5 and 6: The investigations described in thi
- Page 7 and 8: Chapter 1 Chapter 1 Pharmacotherape
- Page 9 and 10: Chapter 1 2.2 Role of calcium Calci
- Page 11 and 12: Chapter 1 The excitation-contractio
- Page 13 and 14: Chapter 1 Antiarrhythmic drugs modi
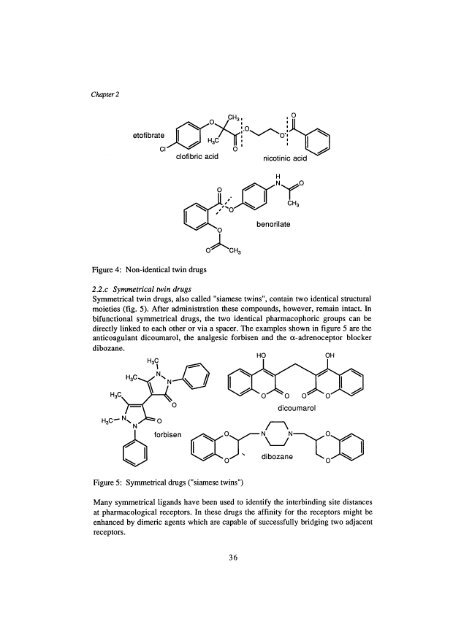
- Page 15 and 16: Chapter 1 Until now, no selective c
- Page 17 and 18: Chapter 1 pyridines are expected to
- Page 19 and 20: Chapter 1 Like the p-adrenoceptor s
- Page 21 and 22: Chapter 1 3.3.3 Phosphodiesterase I
- Page 23 and 24: arteries brain Central Nervous Syst
- Page 25 and 26: Chapter 1 The development of renin
- Page 27 and 28: ^-ADRENOCEPTOR ANTAGONISTS Chapter
- Page 29 and 30: 51 Cromakalim 52 Nicorandil 53 Pina
- Page 31 and 32: Chapter 1 The CCBs are a heterogene
- Page 33 and 34: Chapter 1 4.2 Combination of cardio
- Page 35 and 36: Chapter 1 22 Claremon DA, Baldwin J
- Page 37 and 38: Chapter 1 56 Carini DJ, Chiu AT, Du
- Page 39 and 40: Chapter 2 Chapter 2 Hybrid molecule
- Page 41: identical twin drug (A-A) - 2A non-
- Page 45 and 46: Chapter 2 plasma-protein binding si
- Page 47 and 48: Chapter 2 PAF antagonist, showing o
- Page 49 and 50: Corsano et al. 18 Chapter 2 have sy
- Page 51 and 52: Chapter 2 Görlitzer et al. 21 synt
- Page 53 and 54: Chapter 2 enzyme cyclooxygenase is
- Page 55 and 56: Chapter 2 Table 4: TxA 2/PGH 2 bind
- Page 57 and 58: Chapter 2 the compound had poor ora
- Page 59 and 60: Chapter 2 presented to explain why
- Page 61 and 62: Chapter! When two antihypertensive
- Page 63 and 64: Chapter 2 Table 9: p-Receptor affin
- Page 65 and 66: Chapter 2 dual vasodilating mechani
- Page 67 and 68: Figure 33: Hybrid molecule DG20 (R
- Page 69 and 70: 5.4 Antihypertensive hybrid molecul
- Page 71 and 72: Chapter 2 However, due to the terat
- Page 73 and 74: Figure 43: Hybrid molecule possessi
- Page 75 and 76: Chapter 2 5.4.e Hybrid molecules co
- Page 77 and 78: Chapter 2 The two pairs of enantiom
- Page 79 and 80: I H CH 3 Figure 50: Urapidil, an 04
- Page 81 and 82: Chapter 2 15 Pilar Ortega M, Del Ca
- Page 83 and 84: Chapter 2 AI Morgan TK Jr, Lis R, L
- Page 85 and 86: Chapter 2 68 Baldwin JJ, Lurama WC
- Page 87 and 88: Chapter 3 '2 Chapter 3 Organic nitr
- Page 89 and 90: Chapter 3 phosphorylation of contra
- Page 91 and 92: Chapter 3 In table 1, the in vitro
- Page 93 and 94:
Figure 8: Ligands for the histamine
- Page 95 and 96:
Chapter 3 Nitrate esters can be obt
- Page 97 and 98:
Chapter 3 resulting in the loss of
- Page 99 and 100:
Chapter 3 14 Yeates RA, Possible me
- Page 101 and 102:
Chapter 4 Chapter 4 2 + L-type volt
- Page 103 and 104:
Chapter 4 homologous domains surrou
- Page 105 and 106:
Chapter 4 channel, which is suggest
- Page 107 and 108:
Chapter 4 channel blocking activity
- Page 109 and 110:
Chapter 4 Structural modifications
- Page 111 and 112:
Chapter 4 Table 6: Calcium channel
- Page 113 and 114:
RiOOC COORo compound R\ R 2 nicardi
- Page 115 and 116:
Chapter 4 enantiomer.(-)-S11568 inh
- Page 117 and 118:
Chapter 4 The cardiovascular activi
- Page 119 and 120:
Chapter 4 rat tail artery 73 . Also
- Page 121 and 122:
Chapter 4 The compound HOE 166 18 (
- Page 123 and 124:
Figure 11: Molecular structure of a
- Page 125 and 126:
Chapter 4 SM-6586 was a more potent
- Page 127 and 128:
Chapter 4 inactivated (resting) sta
- Page 129 and 130:
Chapter 4 20 van Amsterdam F.T.M, Z
- Page 131 and 132:
Chapter 4 45 Muto K, Kuroda T, Kawa
- Page 133 and 134:
Chapter 4 67 Gaviraghi G, Lacidipin
- Page 135 and 136:
Chapter 4 95 Goldmann S, Stoltefuß
- Page 137 and 138:
Chapter 5 Synthesis and in vitro ph
- Page 139 and 140:
Chapters
- Page 141 and 142:
Chapters from the 1,4-DHPs 17. In t
- Page 143 and 144:
3 Pharmacology Chapter 5 3.1 In vit
- Page 145 and 146:
Chapter 5 Table II. Calcium blockin
- Page 147 and 148:
Chapter 5 Radioligand binding affin
- Page 149 and 150:
Chapters 13.1 Hz, 2H, pyridine-CHb-
- Page 151 and 152:
2-(2-aminoethylthio)methyl-3,5-dica
- Page 153 and 154:
Chapters 5 Katz AM, In: Calcium ant
- Page 155 and 156:
Chapter 6 Synthesis and in vitro ph
- Page 157 and 158:
Chapter 6 by nifedipine analogues i
- Page 159 and 160:
Chapter 6 4 Results and discussion
- Page 161 and 162:
lipid compartment protein compartme
- Page 163 and 164:
Chapter 6 phthalimide-H), 7.77-7.80
- Page 165 and 166:
Chapter 6 ^-NMR (CDCI3): 1.18 ppm (
- Page 167 and 168:
Chapter 6 *H-NMR (DMSO-d6): 1.07 pp
- Page 169 and 170:
Chapter 7 Chapter 7 Synthesis and i
- Page 171 and 172:
5 Impromidine R 2 9 Arpromidine R 1
- Page 173 and 174:
Chapter 7 generation calcium channe
- Page 175 and 176:
3.2 In vitro histamine Ho-agonistic
- Page 177 and 178:
Chapter 7 As reported previously in
- Page 179 and 180:
Chapter 7 Table III: Relative calci
- Page 181 and 182:
Chapter 7 Figure 4 nicely demonstra
- Page 183 and 184:
Chapter 7 blocking activities are c
- Page 185 and 186:
Chapter 7 4.4 Histamine H ragonisti
- Page 187 and 188:
Chapter 7 chronotropic activity in
- Page 189 and 190:
Chapter 7 N-benzoyl-N'-{3-{[3,5-die
- Page 191 and 192:
Chapter 7 phenyl-// 5), 7.63-7.67 p
- Page 193 and 194:
Chapter 7 and N-C-C-C// 2-imidazole
- Page 195 and 196:
Chapter 7 (s, 1H, pyridine-// 4), 6
- Page 197 and 198:
Chapter 8 Chapter 8 A new series of
- Page 199 and 200:
Chapter 8 structural moiety of impr
- Page 201 and 202:
Chapter 8 All compounds except VUF
- Page 203 and 204:
4 Pharmacology Chapter 8 4.1 Histam
- Page 205 and 206:
Chapter 8 of the 3,3-diphenylpropyl
- Page 207 and 208:
Chapter 8 Based on the observations
- Page 209 and 210:
Chapter 8 iH-NMR (CDCI3): 1.28-1.50
- Page 211 and 212:
Chapter 8 13 Vitali T, Impicciatore
- Page 213 and 214:
Summary The investigations describe
- Page 215 and 216:
Samenvatting Het in dit proefschrif
- Page 217 and 218:
substituenten, zoals een difenylalk
- Page 219 and 220:
Curriculum Vitae Johannes Antonius
- Page 221:
voor de prettige sociale contacten