Kompendium 2021 Forschung & Klinik
Universitätsklinik für Orthopädie und Unfallchirurgie MedUni Wien und AKH Wien; Leitung: o. Univ.-Prof. Dr. Reinhard Windhager www.meduniwien.ac.at/ortho-unfall www.unlimitedmedia.at/orthopaedie2021
Universitätsklinik für Orthopädie und Unfallchirurgie MedUni Wien und AKH Wien; Leitung: o. Univ.-Prof. Dr. Reinhard Windhager
www.meduniwien.ac.at/ortho-unfall
www.unlimitedmedia.at/orthopaedie2021
Erfolgreiche ePaper selbst erstellen
Machen Sie aus Ihren PDF Publikationen ein blätterbares Flipbook mit unserer einzigartigen Google optimierten e-Paper Software.
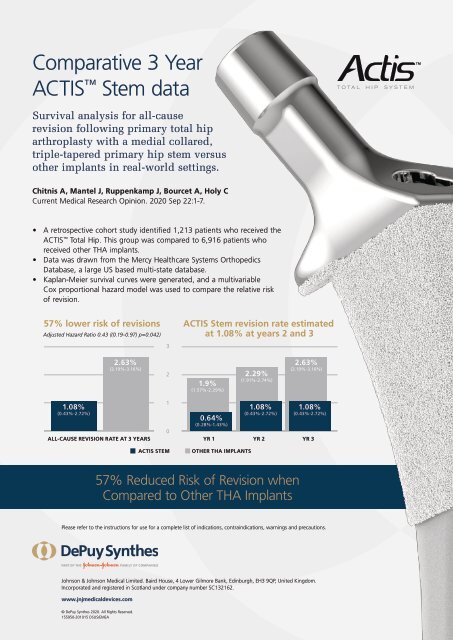
Comparative 3 Year<br />
ACTIS Stem data<br />
Survival analysis for all-cause<br />
revision following primary total hip<br />
arthroplasty with a medial collared,<br />
triple-tapered primary hip stem versus<br />
other implants in real-world settings.<br />
Chitnis A, Mantel J, Ruppenkamp J, Bourcet A, Holy C<br />
Current Medical Research Opinion. 2020 Sep 22:1-7.<br />
• A retrospective cohort study identified 1,213 patients who received the<br />
ACTIS Total Hip. This group was compared to 6,916 patients who<br />
received other THA implants.<br />
• Data was drawn from the Mercy Healthcare Systems Orthopedics<br />
Database, a large US based multi-state database.<br />
• Kaplan-Meier survival curves were generated, and a multivariable<br />
Cox proportional hazard model was used to compare the relative risk<br />
of revision.<br />
57% lower risk of revisions<br />
Adjusted Hazard Ratio 0.43 ((0.19-0.97) p=0.042)<br />
3<br />
ACTIS Stem revision rate estimated<br />
at 1.08% at years 2 and 3<br />
2.63%<br />
(2.19%-3.16%)<br />
2<br />
1.9%<br />
(1.57%-2.29%)<br />
2.29%<br />
(1.91%-2.74%)<br />
2.63%<br />
(2.19%-3.16%)<br />
1.08%<br />
(0.43%-2.72%)<br />
ALL-CAUSE REVISION RATE AT 3 YEARS<br />
1<br />
0<br />
0.64%<br />
(0.28%-1.43%)<br />
1.08%<br />
(0.43%-2.72%)<br />
1.08%<br />
(0.43%-2.72%)<br />
YR 1 YR 2 YR 3<br />
ACTIS STEM<br />
OTHER THA IMPLANTS<br />
57% Reduced Risk of Revision when<br />
Compared to Other THA Implants<br />
Please refer to the instructions for use for a complete list of indications, contraindications, warnings and precautions.<br />
Johnson & Johnson Medical Limited. Baird House, 4 Lower Gilmore Bank, Edinburgh, EH3 9QP, United Kingdom.<br />
Incorporated and registered in Scotland under company number SC132162.<br />
www.jnjmedicaldevices.com<br />
© DePuy Synthes 2020. All Rights Reserved.<br />
155950-201015 DSUS/EMEA