Understanding Clinical Trial Design - Research Advocacy Network
Understanding Clinical Trial Design - Research Advocacy Network
Understanding Clinical Trial Design - Research Advocacy Network
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
UNDERSTANDING CLINICAL TRIAL DESIGN: A TUTORIAL FOR RESEARCH ADVOCATES<br />
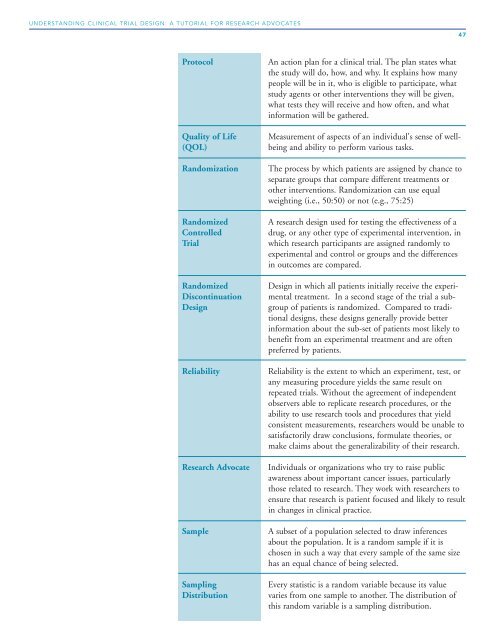
Protocol An action plan for a clinical trial. The plan states what<br />
the study will do, how, and why. It explains how many<br />
people will be in it, who is eligible to participate, what<br />
study agents or other interventions they will be given,<br />
what tests they will receive and how often, and what<br />
information will be gathered.<br />
Quality of Life Measurement of aspects of an individual's sense of well-<br />
(QOL) being and ability to perform various tasks.<br />
Randomization The process by which patients are assigned by chance to<br />
separate groups that compare different treatments or<br />
other interventions. Randomization can use equal<br />
weighting (i.e., 50:50) or not (e.g., 75:25)<br />
Randomized A research design used for testing the effectiveness of a<br />
Controlled drug, or any other type of experimental intervention, in<br />
<strong>Trial</strong> which research participants are assigned randomly to<br />
experimental and control or groups and the differences<br />
in outcomes are compared.<br />
Randomized <strong>Design</strong> in which all patients initially receive the experi-<br />
Discontinuation mental treatment. In a second stage of the trial a sub-<br />
<strong>Design</strong> group of patients is randomized. Compared to traditional<br />
designs, these designs generally provide better<br />
information about the sub-set of patients most likely to<br />
benefit from an experimental treatment and are often<br />
preferred by patients.<br />
Reliability Reliability is the extent to which an experiment, test, or<br />
any measuring procedure yields the same result on<br />
repeated trials. Without the agreement of independent<br />
observers able to replicate research procedures, or the<br />
ability to use research tools and procedures that yield<br />
consistent measurements, researchers would be unable to<br />
satisfactorily draw conclusions, formulate theories, or<br />
make claims about the generalizability of their research.<br />
<strong>Research</strong> Advocate Individuals or organizations who try to raise public<br />
awareness about important cancer issues, particularly<br />
those related to research. They work with researchers to<br />
ensure that research is patient focused and likely to result<br />
in changes in clinical practice.<br />
Sample A subset of a population selected to draw inferences<br />
about the population. It is a random sample if it is<br />
chosen in such a way that every sample of the same size<br />
has an equal chance of being selected.<br />
Sampling Every statistic is a random variable because its value<br />
Distribution varies from one sample to another. The distribution of<br />
this random variable is a sampling distribution.<br />
47