Thermodynamic Quantities for the Ionization Reactions of Buffers
Thermodynamic Quantities for the Ionization Reactions of Buffers
Thermodynamic Quantities for the Ionization Reactions of Buffers
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
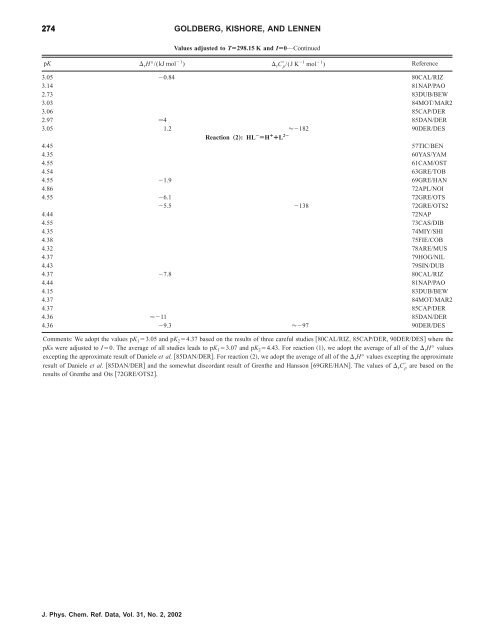
274 GOLDBERG, KISHORE, AND LENNEN<br />
Values adjusted to TÄ298.15 K and IÄ0—Continued<br />
pK rH°/(kJ mol 1 ) rC p /(J K 1 mol 1 ) Reference<br />
3.05 0.84 80CAL/RIZ<br />
3.14 81NAP/PAO<br />
2.73 83DUB/BEW<br />
3.03 84MOT/MAR2<br />
3.06 85CAP/DER<br />
2.97 4 85DAN/DER<br />
3.05 1.2 182 90DER/DES<br />
Reaction „2…: HL À ÄH ¿ ¿L 2À<br />
4.45 57TIC/BEN<br />
4.35 60YAS/YAM<br />
4.55 61CAM/OST<br />
4.54 63GRE/TOB<br />
4.55 1.9 69GRE/HAN<br />
4.86 72APL/NOI<br />
4.55 6.1 72GRE/OTS<br />
5.5 138 72GRE/OTS2<br />
4.44 72NAP<br />
4.55 73CAS/DIB<br />
4.35 74MIY/SHI<br />
4.38 75FIE/COB<br />
4.32 78ARE/MUS<br />
4.37 79HOG/NIL<br />
4.43 79SIN/DUB<br />
4.37 7.8 80CAL/RIZ<br />
4.44 81NAP/PAO<br />
4.15 83DUB/BEW<br />
4.37 84MOT/MAR2<br />
4.37 85CAP/DER<br />
4.36 11 85DAN/DER<br />
4.36 9.3 97 90DER/DES<br />
Comments: We adopt <strong>the</strong> values pK 13.05 and pK 24.37 based on <strong>the</strong> results <strong>of</strong> three careful studies 80CAL/RIZ, 85CAP/DER, 90DER/DES where <strong>the</strong><br />
pKs were adjusted to I0. The average <strong>of</strong> all studies leads to pK 13.07 and pK 24.43. For reaction 1, we adopt <strong>the</strong> average <strong>of</strong> all <strong>of</strong> <strong>the</strong> rH° values<br />
excepting <strong>the</strong> approximate result <strong>of</strong> Daniele et al. 85DAN/DER. For reaction 2, we adopt <strong>the</strong> average <strong>of</strong> all <strong>of</strong> <strong>the</strong> rH° values excepting <strong>the</strong> approximate<br />
result <strong>of</strong> Daniele et al. 85DAN/DER and <strong>the</strong> somewhat discordant result <strong>of</strong> Gren<strong>the</strong> and Hansson 69GRE/HAN. The values <strong>of</strong> rC p are based on <strong>the</strong><br />
results <strong>of</strong> Gren<strong>the</strong> and Ots 72GRE/OTS2.<br />
J. Phys. Chem. Ref. Data, Vol. 31, No. 2, 2002