Antiemetics for reducing vomiting related to acute ... - Update Software
Antiemetics for reducing vomiting related to acute ... - Update Software
Antiemetics for reducing vomiting related to acute ... - Update Software
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
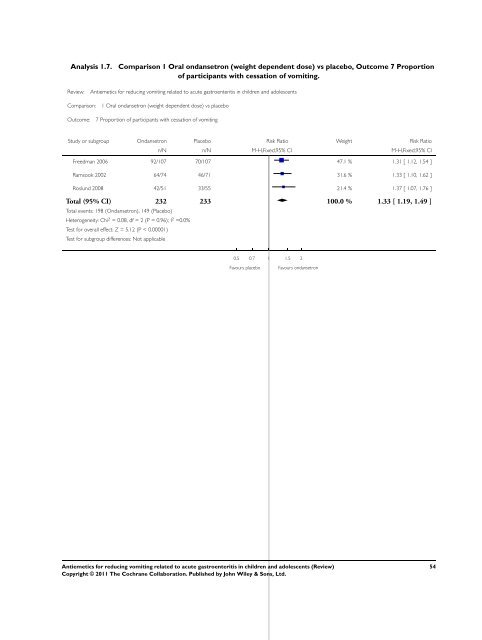
Analysis 1.7. Comparison 1 Oral ondansetron (weight dependent dose) vs placebo, Outcome 7 Proportion<br />
of participants with cessation of <strong>vomiting</strong>.<br />
Review: <strong>Antiemetics</strong> <strong>for</strong> <strong>reducing</strong> <strong>vomiting</strong> <strong>related</strong> <strong>to</strong> <strong>acute</strong> gastroenteritis in children and adolescents<br />
Comparison: 1 Oral ondansetron (weight dependent dose) vs placebo<br />
Outcome: 7 Proportion of participants with cessation of <strong>vomiting</strong><br />
Study or subgroup Ondansetron Placebo Risk Ratio Weight Risk Ratio<br />
n/N n/N M-H,Fixed,95% CI M-H,Fixed,95% CI<br />
Freedman 2006 92/107 70/107 47.1 % 1.31 [ 1.12, 1.54 ]<br />
Ramsook 2002 64/74 46/71 31.6 % 1.33 [ 1.10, 1.62 ]<br />
Roslund 2008 42/51 33/55 21.4 % 1.37 [ 1.07, 1.76 ]<br />
Total (95% CI) 232 233 100.0 % 1.33 [ 1.19, 1.49 ]<br />
Total events: 198 (Ondansetron), 149 (Placebo)<br />
Heterogeneity: Chi 2 = 0.08, df = 2 (P = 0.96); I 2 =0.0%<br />
Test <strong>for</strong> overall effect: Z = 5.12 (P < 0.00001)<br />
Test <strong>for</strong> subgroup differences: Not applicable<br />
0.5 0.7 1 1.5 2<br />
Favours placebo Favours ondansetron<br />
<strong>Antiemetics</strong> <strong>for</strong> <strong>reducing</strong> <strong>vomiting</strong> <strong>related</strong> <strong>to</strong> <strong>acute</strong> gastroenteritis in children and adolescents (Review)<br />
Copyright © 2011 The Cochrane Collaboration. Published by John Wiley & Sons, Ltd.<br />
54