The report is available in English with a French summary - KCE
The report is available in English with a French summary - KCE
The report is available in English with a French summary - KCE
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
70 Plasma <strong>KCE</strong> Reports 120<br />
Chronic fatigue syndrome<br />
<strong>The</strong> Chronic fatigue syndrome (CFS) <strong>is</strong> a condition character<strong>is</strong>ed by severe, d<strong>is</strong>abl<strong>in</strong>g<br />
fatigue <strong>in</strong> the absence of exertion, and <strong>is</strong> marked by a dramatic decl<strong>in</strong>e <strong>in</strong> activity level.<br />
Currently, the aetiology of CFS rema<strong>in</strong>s unknown.<br />
<strong>The</strong> beneficial effect of IG for CFS patients <strong>is</strong> controversial. Three RCTs obta<strong>in</strong>ed mixed<br />
results: a RCT conducted <strong>in</strong> 1990 found improvements <strong>in</strong> symptom scores and<br />
functional capacity at three months; 143 a second found improvement <strong>in</strong> immune<br />
<strong>in</strong>dicators but not <strong>in</strong> symptom and functional measures, 144 the third trial was more<br />
recent and larger (<strong>in</strong>volv<strong>in</strong>g 99 adult patients). 145 It found no effect of treatment, but<br />
<strong>report</strong>ed adverse reactions <strong>in</strong> 70-80% patients, <strong>with</strong> no relationship to IG treatment. It<br />
concluded that IG cannot be recommended as a therapy for CFS.<br />
<strong>The</strong>re <strong>is</strong> conflict<strong>in</strong>g evidence on the effect of IG <strong>in</strong> CFS, but the larger and more<br />
recent trial showed no effect, <strong>with</strong> a high rate of adverse events.<br />
3.3 CONSUMPTION OF IMMUNOGLOBULINS<br />
3.3.1 Consumption <strong>in</strong> other countries<br />
Few studies <strong>in</strong>vestigat<strong>in</strong>g IG use and its trends are <strong>available</strong>. No study has been found on<br />
IG consumption <strong>in</strong> European countries. However, eight studies <strong>with</strong> quantitative data<br />
on recent IG use per <strong>in</strong>dication were found from Australia, Canada, New Zealand and<br />
the US. 146,147,148,149,150,151,152,153 .<br />
3.3.1.1 Consumption by medical speciality and <strong>in</strong>dication<br />
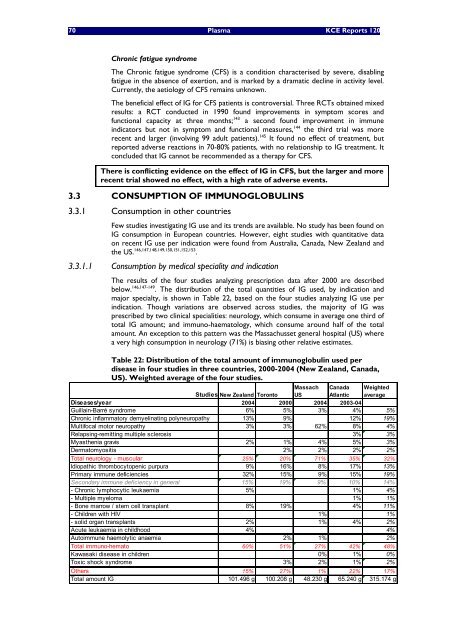
<strong>The</strong> results of the four studies analyz<strong>in</strong>g prescription data after 2000 are described<br />
below. 146,147-149 . <strong>The</strong> d<strong>is</strong>tribution of the total quantities of IG used, by <strong>in</strong>dication and<br />
major specialty, <strong>is</strong> shown <strong>in</strong> Table 22, based on the four studies analyz<strong>in</strong>g IG use per<br />
<strong>in</strong>dication. Though variations are observed across studies, the majority of IG was<br />
prescribed by two cl<strong>in</strong>ical specialities: neurology, which consume <strong>in</strong> average one third of<br />
total IG amount; and immuno-haematology, which consume around half of the total<br />
amount. An exception to th<strong>is</strong> pattern was the Massachusset general hospital (US) where<br />
a very high consumption <strong>in</strong> neurology (71%) <strong>is</strong> bias<strong>in</strong>g other relative estimates.<br />
Table 22: D<strong>is</strong>tribution of the total amount of immunoglobul<strong>in</strong> used per<br />
d<strong>is</strong>ease <strong>in</strong> four studies <strong>in</strong> three countries, 2000-2004 (New Zealand, Canada,<br />
US). Weighted average of the four studies.<br />
Studies New Zealand Toronto<br />
Massach<br />
US<br />
Canada<br />
Atlantic<br />
Weighted<br />
average<br />
D<strong>is</strong>eases/year 2004 2000 2004 2003-04<br />
Guilla<strong>in</strong>-Barré syndrome 6% 5% 3% 4% 5%<br />
Chronic <strong>in</strong>flammatory demyel<strong>in</strong>at<strong>in</strong>g polyneuropathy 13% 9% 12% 19%<br />
Multifocal motor neuropathy 3% 3% 62%<br />
8% 4%<br />
Relaps<strong>in</strong>g-remitt<strong>in</strong>g multiple scleros<strong>is</strong> 3% 3%<br />
Myasthenia grav<strong>is</strong> 2% 1% 4% 5% 3%<br />
Dermatomyosit<strong>is</strong> 2% 2% 2% 2%<br />
Total neurology - muscular 25% 20% 71% 35% 32%<br />
Idiopathic thrombocytopenic purpura 9% 16% 8% 17% 13%<br />
Primary immune deficiencies 32% 15% 9% 15% 19%<br />
Secondary immune deficiency <strong>in</strong> general 15% 19% 9% 10% 14%<br />
- Chronic lymphocytic leukaemia 5% 1% 4%<br />
- Multiple myeloma 1% 1%<br />
- Bone marrow / stem cell transplant 8% 19% 4% 11%<br />
- Children <strong>with</strong> HIV 1% 1%<br />
- solid organ transplants 2% 1% 4% 2%<br />
Acute leukaemia <strong>in</strong> childhood 4% 4%<br />
Autoimmune haemolytic anaemia 2% 1% 2%<br />
Total immuno-hemato 60% 51% 27% 42% 48%<br />
Kawasaki d<strong>is</strong>ease <strong>in</strong> children 0% 1% 0%<br />
Toxic shock syndrome 3% 2% 1% 2%<br />
Others 15% 27% 1% 22% 17%<br />
Total amount IG 101.496 g 100.208 g 48.230 g 65.240 g 315.174 g