WOC 6e Guide to Microscopy
WOC 6e Guide to Microscopy
WOC 6e Guide to Microscopy
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
E-face<br />
P-face<br />
0.1 m<br />
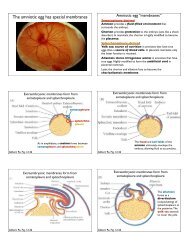
Figure A-35 Freeze Fracturing of the Plasma Membrane. This<br />
electron micrograph shows the exposed faces of the plasma<br />
membranes of two adjacent endocrine cells from a rat pancreas as<br />
revealed by freeze fracturing. The P face is the inner surface of the<br />
lipid monolayer on the pro<strong>to</strong>plasmic side of the plasma membrane.<br />
The E face is the inner surface of the lipid monolayer on the<br />
exterior side of the plasma membrane. The P face is much<br />
more richly studded with intramembranous particles than the E<br />
face. The arrows indicate the “step” along which the fracture<br />
plane passed from the interior of the plasma membrane of one<br />
cell <strong>to</strong> the interior of the plasma membrane of a neighboring cell.<br />
The step therefore represents the thickness of the intercellular<br />
space (TEM).<br />
especially useful in exploring the cy<strong>to</strong>skele<strong>to</strong>n and examining<br />
its connections with other structures of the cell.<br />
STEREO ELECTRON MICROSCOPY ALLOWS SPECIMENS<br />
TO BE VIEWED IN THREE DIMENSIONS<br />
Electron microscopists frequently want <strong>to</strong> visualize specimens<br />
in three dimensions. Shadowing, freeze fracturing, and<br />
scanning electron microscopy are useful for this purpose, as<br />
is another specialized technique called stereo electron<br />
microscopy. In stereo electron microscopy, three-dimensional<br />
information is obtained by pho<strong>to</strong>graphing the same specimen<br />
at two slightly different angles. This is accomplished using a<br />
special specimen stage that can be tilted relative <strong>to</strong> the electron<br />
beam. The specimen is first tilted in one direction and pho<strong>to</strong>graphed,<br />
then tilted an equal amount in the opposite direction<br />
and pho<strong>to</strong>graphed again.<br />
The two micrographs are then mounted side by side as<br />
a stereo pair. When you view a stereo pair through a stereoscopic<br />
viewer, your brain uses the two independent images<br />
<strong>to</strong> construct a three-dimensional view that gives a striking<br />
sense of depth <strong>to</strong> the structure under investigation. Figure<br />
A-26 Appendix Principles and Techniques of <strong>Microscopy</strong><br />
0.5 m<br />
Figure A-36 Stereo Electron <strong>Microscopy</strong>. The polytene chromosome<br />
seen in this high-voltage electron micrograph is shown here<br />
as a stereo pair of pho<strong>to</strong>s that can be fused optically <strong>to</strong> generate a<br />
three-dimensional image. The two pho<strong>to</strong>graphs were taken by<br />
tilting the specimen stage first 5° <strong>to</strong> the right, then 5° <strong>to</strong> the left of<br />
the electron beam. For a three-dimensional view, a stereo pair can<br />
be examined with a stereoscopic viewer. Alternatively, simply let<br />
your eyes cross slightly, fusing the two micrographs in<strong>to</strong> a single<br />
image (HVEM).<br />
A-36 is a stereo pair of a Drosophila polytene chromosome<br />
imaged by high-voltage electron microscopy. Using a stereo<br />
viewer or allowing your eyes <strong>to</strong> fuse the two images visually<br />
creates a striking, three-dimensional view of the chromosome.<br />
SPECIMEN PREPARATION FOR SCANNING ELECTRON<br />
MICROSCOPY INVOLVES FIXATION BUT NOT<br />
SECTIONING<br />
When preparing a specimen for scanning electron<br />
microscopy, the goal is <strong>to</strong> preserve the structural features of<br />
the cell surface and <strong>to</strong> treat the tissue in a way that minimizes<br />
damage by the electron beam. The procedure is actually similar<br />
<strong>to</strong> the preparation of ultrathin sections for transmission<br />
electron microscopy, but without the sectioning step. The<br />
tissue is fixed in aldehyde, postfixed in osmium tetroxide, and<br />
dehydrated by processing through a series of alcohol solutions.<br />
The tissue is then placed in a fluid such as liquid carbon<br />
dioxide in a heavy metal canister called a critical point<br />
dryer, which is used <strong>to</strong> dry the specimen under conditions<br />
of controlled temperature and pressure. This helps keep