School of Engineering and Science - Jacobs University
School of Engineering and Science - Jacobs University
School of Engineering and Science - Jacobs University
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
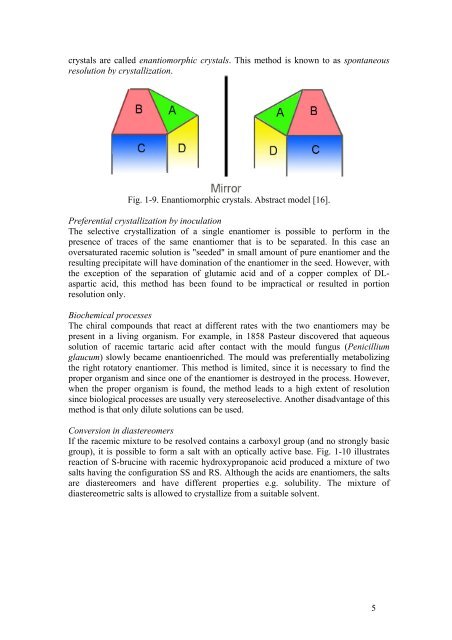
crystals are called enantiomorphic crystals. This method is known to as spontaneous<br />
resolution by crystallization.<br />
Fig. 1-9. Enantiomorphic crystals. Abstract model [16].<br />
Preferential crystallization by inoculation<br />
The selective crystallization <strong>of</strong> a single enantiomer is possible to perform in the<br />
presence <strong>of</strong> traces <strong>of</strong> the same enantiomer that is to be separated. In this case an<br />
oversaturated racemic solution is "seeded" in small amount <strong>of</strong> pure enantiomer <strong>and</strong> the<br />
resulting precipitate will have domination <strong>of</strong> the enantiomer in the seed. However, with<br />
the exception <strong>of</strong> the separation <strong>of</strong> glutamic acid <strong>and</strong> <strong>of</strong> a copper complex <strong>of</strong> DLaspartic<br />
acid, this method has been found to be impractical or resulted in portion<br />
resolution only.<br />
Biochemical processes<br />
The chiral compounds that react at different rates with the two enantiomers may be<br />
present in a living organism. For example, in 1858 Pasteur discovered that aqueous<br />
solution <strong>of</strong> racemic tartaric acid after contact with the mould fungus (Penicillium<br />
glaucum) slowly became enantioenriched. The mould was preferentially metabolizing<br />
the right rotatory enantiomer. This method is limited, since it is necessary to find the<br />
proper organism <strong>and</strong> since one <strong>of</strong> the enantiomer is destroyed in the process. However,<br />
when the proper organism is found, the method leads to a high extent <strong>of</strong> resolution<br />
since biological processes are usually very stereoselective. Another disadvantage <strong>of</strong> this<br />
method is that only dilute solutions can be used.<br />
Conversion in diastereomers<br />
If the racemic mixture to be resolved contains a carboxyl group (<strong>and</strong> no strongly basic<br />
group), it is possible to form a salt with an optically active base. Fig. 1-10 illustrates<br />
reaction <strong>of</strong> S-brucine with racemic hydroxypropanoic acid produced a mixture <strong>of</strong> two<br />
salts having the configuration SS <strong>and</strong> RS. Although the acids are enantiomers, the salts<br />
are diastereomers <strong>and</strong> have different properties e.g. solubility. The mixture <strong>of</strong><br />
diastereometric salts is allowed to crystallize from a suitable solvent.<br />
5