A guide for the preparation and use of buffers in biological systems
A guide for the preparation and use of buffers in biological systems
A guide for the preparation and use of buffers in biological systems
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
Commonly Used Buffer Media <strong>in</strong> Biological Research<br />
Krebs-Henseleit bicarbonate buffer, pH 7.4<br />
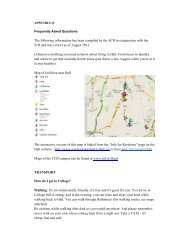
119 mM NaCl<br />
4.7 mM KCl<br />
2.5 mM CaCl 2<br />
1.2 mM MgSO 4<br />
1.2 mM KH 2<br />
PO 4<br />
25 mM NaHCO 3<br />
pH 7.4 (at 37°C) when equilibrated with 95% O 2<br />
<strong>and</strong> 5% CO 2<br />
. Adjust <strong>the</strong> pH<br />
be<strong>for</strong>e <strong>use</strong>.<br />
Hank’s Biocarbonate Buffer, pH 7.4<br />
137 mM NaCl<br />
5.4 mM KCl<br />
0.25 mM Na 2<br />
HPO 4<br />
0.44 mM KH 2<br />
PO 4<br />
1.3 mM CaCl 2<br />
1.0 mM MgSO 4<br />
4.2 mM NaHCO 3<br />
pH 7.4 (at 37°C) when equilibrated with 95% O 2<br />
<strong>and</strong> 5% CO 2<br />
. Adjust <strong>the</strong> pH<br />
be<strong>for</strong>e <strong>use</strong>.<br />
Phosphate Buffered Sal<strong>in</strong>e (PBS), pH 7.4<br />
150 mM NaCl<br />
10 mM Potassium Phosphate buffer<br />
(1 liter PBS can be prepared by dissolv<strong>in</strong>g 8.7 g NaCl, 1.82 g<br />
K 2<br />
HPO 4<br />
.3H 2<br />
O, <strong>and</strong> 0.23 g KH 2<br />
PO 4<br />
<strong>in</strong> 1 liter <strong>of</strong> distilled water.<br />
Adjust <strong>the</strong> pH be<strong>for</strong>e <strong>use</strong>).<br />
A variation <strong>of</strong> PBS can also be prepared as follows:<br />
137 mM NaCl<br />
2.7 mM KCl<br />
10 mM Na 2<br />
HPO 4<br />
1.76 mM KH 2<br />
PO 4<br />
22