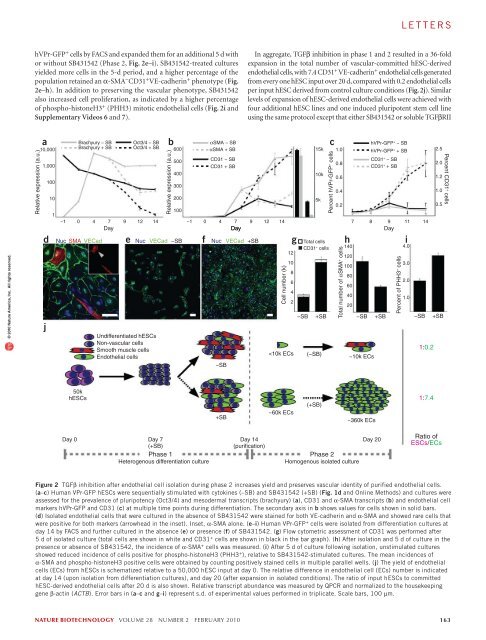
lettersa1.5 kb hVPrIntron 112.5 kbATGbhVPr-GFPc10 510 4LTRExon 164 bpExon 2226 bpα-CD3110 3dU3 U5 GA RRE eGFP WPRE U3U5SuspensionAdherent10 20−10 2−100−10 2 010 2−203Day (−1) 0 1 247 1410 3 10 4α-VEGFR2Isolation10 5BMP-4Activin AFGF-2Phase 1differentiationMesodermal and vascular specificationVEGF-ASB431542EC amplificationPhase 2isolation© 2010 Nature America, Inc. All rights reserved.ehVPr-GFPfNuc VECad hVPr-GFPPropidium iodide10 310 210 1g−SBh+SB10 0 0.15% 1.8%10 0 10 1 10 2 10 3 10 0 10 1 10 2 10 3hVPr-GFPi% hVPr-GFP + cells2.01.61.20.80.4Day 14Grouped IsolatedP < .001 P < .01d0 + N d7 + N d7 +Figure 1 Sequential TGFβ activation followed by inhibition during phase 1 differentiation promotes a tenfold expansion of hVPr-GFP + hESC-derived cells.(a) A 1.5-kb fragment of the putative human VE-cadherin promoter (hVPr) region was isolated from a BAC clone and placed upstream of GFP in a lentiviralexpression vector (hVPr-GFP). (b) Spontaneously differentiating embryoid bodies exhibited expression of hVPr-GFP in tubular structures. Inset, merge of GFPand brightfield views. (c) Flow cytometric analysis showed hVPr-GFP + cells were positive for the vascular markers CD31 and VEGFR2. (d) Schematic diagramshowing the sequence in which BMP4, activinA, FGF-2, VEGF-A and SB431542 were added and removed from differentiation cultures. EC, endothelial cell.(e) Adherent hVPr-GFP cultures stimulated with SB431542 (10 µM) resulted in differentiation of hESCs into monolayers of hVPr-GFP + adherent cells.Inset, hVPr-GFP + cells alone. (f) Human VPr-GFP + cells were immuno-positive for VE-cadherin. Blue, nuclear counterstain. Inset, hVPr-GFP + cells alone.(g,h) The proportion of hVPr-GFP + cells was measured by flow cytometry at day 14 after culture in the absence (–SB; g) and presence (+SB; h) of SB431542.(i) Measurement of hVPr-GFP + cells at day 14 when embryoid bodies were cultured either in groups or as isolated embryoid bodies and SB431542 was addedat day 0, day 7 or not at all (N). Error bars represent s.d. of experimental values performed in triplicate. Scale bars, 100 µm.(data not shown). We therefore screened for bioactive small moleculesthat would improve vascular differentiation. Screening of >20 moleculesassociated with early developmental signaling pathways (SupplementaryTable 1) showed that the TGFβ-inhibitory molecule SB431542 (ref. 11)reproducibly increased the yield of hVPr-GFP + cells. Adding SB431542to differentiation cultures at day 7 resulted in the formation of hVPr-GFP + VE-cadherin + monolayers (Fig. 1e,f), which, upon dissociation,yielded tenfold more hVPr-GFP + endothelial cells than cultures stimulatedby cytokines alone (Fig. 1g–i). No hVPr-GFP + cells were generatedif SB431542 was added at the onset of differentiation (day 0), suggestingthat vascular commitment depends on active TGFβ/activin/nodal signalingbefore day 7.Kinetic analysis of differentiation suggested a shift from a pluripotentphenotype (Oct3/4 + ; Fig. 2a) to a vascular phenotype (CD31 + ;Fig. 2b,c) through a mesodermal intermediate (brachyury + ; Fig. 2a).Addition of SB431542 to differentiating hESC cultures at day 7 acceleratedthe reduction of Oct3/4 and brachyury and increased the numberof hVPr-GFP + CD31 + cells beginning at about day 9, while reducingexpression of α-SMA (Fig. 2b,c). After isolation from heterogenouscultures by FACS, endothelial cells grown in the absence of TGFβ inhibitionretained high expression of CD31 but also expressed α-SMA(Supplementary Video 5), indicating that these endothelial cell–likecells had not assumed a terminally committed vascular fate.Expression of α-SMA in hESC-derived endothelial cells suggesteda degree of plasticity that is not present in terminally differentiatedendothelial cells (human umbilical vein endothelial cells; HUVEC,Fig. 2b). Indeed, extended culture (>10 d after FACS isolation) of hESCderivedendothelial cells in the absence of TGFβ inhibition yielded asubstantial number of cells co-expressing VE-cadherin and α-SMA(Fig. 2d). One explanation for the increased percentage of endothelialcells in SB431542-stimulated cultures is maintenance of the vascularcommittedstate after specification. To test the capacity of TGFβ inhibitionto promote expansion of pure populations of hESC-derivedendothelial cells, we dissociated day 14 differentiation cultures, isolated162 volume 28 number 2 february 2010 nature biotechnology
lettershVPr-GFP + cells by FACS and expanded them for an additional 5 d withor without SB431542 (Phase 2, Fig. 2e–i). SB431542-treated culturesyielded more cells in the 5-d period, and a higher percentage of thepopulation retained an α-SMA − CD31 + VE-cadherin + phenotype (Fig.2e–h). In addition to preserving the vascular phenotype, SB431542also increased cell proliferation, as indicated by a higher percentageof phospho-histoneH3 + (PHH3) mitotic endothelial cells (Fig. 2i andSupplementary Videos 6 and 7).In aggregate, TGFβ inhibition in phase 1 and 2 resulted in a 36-foldexpansion in the total number of vascular-committed hESC-derivedendothelial cells, with 7.4 CD31 + VE-cadherin + endothelial cells generatedfrom every one hESC input over 20 d, compared with 0.2 endothelial cellsper input hESC derived from control culture conditions (Fig. 2j). Similarlevels of expansion of hESC-derived endothelial cells were achieved withfour additional hESC lines and one induced pluripotent stem cell lineusing the same protocol except that either SB431542 or soluble TGFβRII© 2010 Nature America, Inc. All rights reserved.Relative expression (a.u.)a10,0001,000dj100101Brachyury − SBBrachyury + SBNuc SMA VECadeOct3/4 − SBOct3/4 + SBNucUndifferentiated hESCsNon-vascular cellsSmooth muscle cellsEndothelial cellsbRelative expression (a.u.)600500400300200100VECad −SBfαSMA − SBαSMA + SBCD31 − SBCD31 + SBNuc−SBVECad+SBCell number (k)
- Page 3 and 4:
volume 28 number 2 february 2010COM
- Page 5 and 6:
in this issue© 2010 Nature America
- Page 7 and 8:
© 2010 Nature America, Inc. All ri
- Page 10 and 11:
NEWS© 2010 Nature America, Inc. Al
- Page 12 and 13:
NEWS© 2010 Nature America, Inc. Al
- Page 14 and 15:
NEWS© 2010 Nature America, Inc. Al
- Page 16 and 17:
© 2010 Nature America, Inc. All ri
- Page 18 and 19: © 2010 Nature America, Inc. All ri
- Page 20 and 21: © 2010 Nature America, Inc. All ri
- Page 22 and 23: NEWS feature© 2010 Nature America,
- Page 24 and 25: uilding a businessComing to termsDa
- Page 26 and 27: uilding a business© 2010 Nature Am
- Page 28 and 29: correspondence© 2010 Nature Americ
- Page 30 and 31: correspondence© 2010 Nature Americ
- Page 32 and 33: correspondence© 2010 Nature Americ
- Page 34 and 35: correspondence© 2010 Nature Americ
- Page 36 and 37: case studyNever againcommentaryChri
- Page 38 and 39: COMMENTARY© 2010 Nature America, I
- Page 40 and 41: COMMENTARY© 2010 Nature America, I
- Page 42 and 43: patents© 2010 Nature America, Inc.
- Page 44 and 45: patents© 2010 Nature America, Inc.
- Page 46 and 47: news and viewsChIPs and regulatory
- Page 48 and 49: news and viewsFrom genomics to crop
- Page 50 and 51: news and views© 2010 Nature Americ
- Page 52 and 53: news and views© 2010 Nature Americ
- Page 54 and 55: e s o u r c eRational association o
- Page 56 and 57: e s o u r c e© 2010 Nature America
- Page 58 and 59: e s o u r c e© 2010 Nature America
- Page 60 and 61: e s o u r c e© 2010 Nature America
- Page 62 and 63: © 2010 Nature America, Inc. All ri
- Page 64 and 65: B r i e f c o m m u n i c at i o n
- Page 66 and 67: i e f c o m m u n i c at i o n sAUT
- Page 70 and 71: letters© 2010 Nature America, Inc.
- Page 72 and 73: letters© 2010 Nature America, Inc.
- Page 74 and 75: l e t t e r sReal-time imaging of h
- Page 76 and 77: l e t t e r sFigure 2 Time-lapse li
- Page 78 and 79: l e t t e r s© 2010 Nature America
- Page 80 and 81: l e t t e r sRational design of cat
- Page 82 and 83: l e t t e r s© 2010 Nature America
- Page 84 and 85: l e t t e r s© 2010 Nature America
- Page 86 and 87: sample fluorescence was measured as
- Page 88 and 89: careers and recruitmentFourth quart