336 BEATA PASKER et al.inhibitory on bitterness and may be used for practicalpurposes to inhibit the bitterness of food anddrugs (11, 12). PA was also used to stabilize liposomesencapsulating an emulsion nanodroplets usedfor ultrasonically activated drug delivery (13). Anumber of studies have revealed a decreasing toxicityof amphotericin B administered in the vesicles,liposomes, micelles and emulsions based on negativecharged phospholipids, like PA and phosphatidylglycerol(14, 15). PA monosodium salt wasalso examined as an additive in cationic arginineglyceride conjugates which were used as cationicsurf<strong>acta</strong>nts (16). PA can be easily prepared by phospholipidshydrolysis using plant extracts with phospholipaseD (PLD). PLD was first isolated from carrot(17), is widespread in plants, animals andmicroorganisms and is well known for hydrolyticand transphosphatidylation activity towards naturalphospholipids and their synthetic analogs. PLD catalyzeshydrolysis of the terminal phosphodiesterbond on phospholipid giving PA and free aminoalcoholgroup. In the presence of an another alcoholicnucleophile, PLD catalyzes exchanging the polarhead group, giving a new phosphoester bond (18-20). Many previous studies, using crude and purified,free or immobilized PLD from plant and microbial,showed that this enzyme is capable ofhydrolyzing a broad range of phospholipids.However, significant differences of the substrate forPLD may occur, PC is assumed as the best preferablesubstrate for this enzyme. It is well known thatPLD activity depends on source and purity ofenzyme, structure and form of phospholipid, concentrationof Ca 2+ and pH level. The most importantdifference in phospholipid structure (from differentsources) is their fatty acids profile, especiallylength and degree of unsaturation of carbon chain.It is known from previous studies on egg, soybeanand synthetic phospholipids that the degree of fattyacids saturation in phospholipids have influence onthe PLD activity. PLD from cabbage andStreptomyces sp. are exploited for a number ofphospholipid-transforming reactions in the laboratoryas well as on industrial scale (21-23).Phospholipids from soya and egg yolk are used as asource of natural PA. Although hydrolytic activityof PLD from different sources in hydrolysis of egg,soybean and synthetic phospholipids is welldescribed in many studies, activity of cabbage PLDextracts in hydrolysis of rapeseed phospholipids hasnot been reported. Lower sensitivity to oxidation ofrapeseed phospholipids compared to soya ones givea chance to use them as a more stable phospholipidsderivatives. Furthermore, there is no standards forrapeseed PA, as yet. For that reasons, in our studythe hydrolysis of rapeseed phosphatidylcholine(RPC) to rapeseed PA (RPA) in a biphasic systemwas examined. Purified by us PLD extracts, fromthree varieties of cabbage were used and the resultswere compared with standard cabbage phospholipase(PLD st ).MATERIALS AND METHODSEnzymes and chemicalsPLD st from cabbage (type IV, activity 335units/mg solid, liberated 1 µmol/h choline from eggat pH 6.5 and temp. 30 O C) and egg PA sodium salt(purity 99%) were from Sigma-Aldrich (St. Louis,MO, USA). Solvents of HPLC grade and TLC silicagel G 60 plates were purchased from Merck(Darmstadt, Germany). All other solvents andreagents of analytical grade were from POCH(Gliwice, Poland).Preparation of rapeseed phosphatidylcholineCrude commercial rapeseed lecithin, free oferucic acid and glucosinolates (00-type rapeseed),was obtained from the Company Kruszwica SA(Brzeg, Poland). The raw material was deoiled withacetone and fractionated with 95% ethanol, to obtainrapeseed lecithin ethanol soluble fraction, by themethod described elsewhere (24). Further purificationof lecithin ethanol soluble fraction was performedby column chromatography on aluminumoxide as previously described (25). As a result theRPC with 99% purity (HPLC) was obtained.Preparation of phospholipase D cabbage extractsFresh cabbage varieties: savoy cabbage(Brassica oleracea var. sabauda), white cabbage(Brassica oleracea var. capitata f. alba) and brusselssprouts (Brassica oleracea var. gemmifera),were obtained from the local market. Crude PLDextracts were prepared by the method of Davidsonand Long (26). Hundred grams of cabbage leaves orsprouts were homogenized with 200 mL of water at4 O C for 5 min at 4000 rpm (MPV 120 Homogenizer,MPV MED Instruments, Poland). Homogenatewas stored at 4 O C for 60 min and filtered.After that, the homogenate was centrifuged twice at4000 rpm for 15 min at 4 O C (MPV 325 centrifugeMPV MED Instruments, Poland). The supernatantwas heated at 50 O C for 5 min, immediately cooled,and centrifuged as described. Finally PLD extractswere freeze-dried at -50 O C, 0.03 mBar (FreezoneVI, Labconco, USA) to yield a stable pale beigepowder.
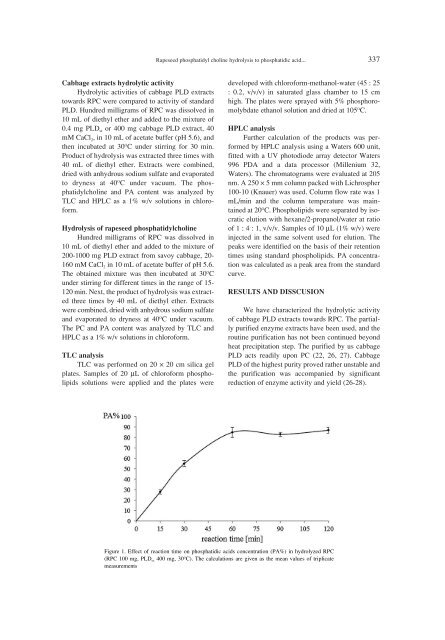
Rapeseed phosphatidyl choline hydrolysis to phosphatidic acid... 337Cabbage extracts hydrolytic activityHydrolytic activities of cabbage PLD extractstowards RPC were compared to activity of standardPLD. Hundred milligrams of RPC was dissolved in10 mL of diethyl ether and added to the mixture of0.4 mg PLD st or 400 mg cabbage PLD extract, 40mM CaCl 2 , in 10 mL of acetate buffer (pH 5.6), andthen incubated at 30 O C under stirring for 30 min.Product of hydrolysis was extracted three times with40 mL of diethyl ether. Extracts were combined,dried with anhydrous sodium sulfate and evaporatedto dryness at 40 O C under vacuum. The phosphatidylcholineand PA content was analyzed byTLC and HPLC as a 1% w/v solutions in chloroform.Hydrolysis of rapeseed phosphatidylcholineHundred milligrams of RPC was dissolved in10 mL of diethyl ether and added to the mixture of200-1000 mg PLD extract from savoy cabbage, 20-160 mM CaCl 2 in 10 mL of acetate buffer of pH 5.6.The obtained mixture was then incubated at 30 O Cunder stirring for different times in the range of 15-120 min. Next, the product of hydrolysis was extractedthree times by 40 mL of diethyl ether. Extractswere combined, dried with anhydrous sodium sulfateand evaporated to dryness at 40 O C under vacuum.The PC and PA content was analyzed by TLC andHPLC as a 1% w/v solutions in chloroform.TLC analysisTLC was performed on 20 ◊ 20 cm silica gelplates. Samples of 20 µL of chloroform phospholipidssolutions were applied and the plates weredeveloped with chloroform-methanol-water (45 : 25: 0.2, v/v/v) in saturated glass chamber to 15 cmhigh. The plates were sprayed with 5% phosphoromolybdateethanol solution and dried at 105 O C.HPLC analysisFurther calculation of the products was performedby HPLC analysis using a Waters 600 unit,fitted with a UV photodiode array detector Waters996 PDA and a data processor (Millenium 32,Waters). The chromatograms were evaluated at 205nm. A 250 ◊ 5 mm column packed with Lichrospher100-10 (Knauer) was used. Column flow rate was 1mL/min and the column temperature was maintainedat 20 O C. Phospholipids were separated by isocraticelution with hexane/2-propanol/water at ratioof 1 : 4 : 1, v/v/v. Samples of 10 µL (1% w/v) wereinjected in the same solvent used for elution. Thepeaks were identified on the basis of their retentiontimes using standard phospholipids. PA concentrationwas calculated as a peak area from the standardcurve.RESULTS AND DISSCUSIONWe have characterized the hydrolytic activityof cabbage PLD extracts towards RPC. The partiallypurified enzyme extracts have been used, and theroutine purification has not been continued beyondheat precipitation step. The purified by us cabbagePLD acts readily upon PC (22, 26, 27). CabbagePLD of the highest purity proved rather unstable andthe purification was accompanied by significantreduction of enzyme activity and yield (26-28).Figure 1. Effect of reaction time on phosphatidic acids concentration (PA%) in hydrolyzed RPC(RPC 100 mg, PLD sc 400 mg, 30 O C). The calculations are given as the mean values of triplicatemeasurements