Staff Members of the Institute of Biochemistry, TU - Institut für ...
Staff Members of the Institute of Biochemistry, TU - Institut für ...
Staff Members of the Institute of Biochemistry, TU - Institut für ...
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
Storage <strong>of</strong> non-polar lipids in lipid droplets and mobilization<br />
Yeast cells like most o<strong>the</strong>r cell types have <strong>the</strong> capacity to store non-polar lipids. In <strong>the</strong> case <strong>of</strong><br />
Saccharomyces cerevisiae triacylglycerols (TG) and steryl esters (SE) are <strong>the</strong> predominant<br />
lipid storage molecules which accumulate in subcellular structures named lipid<br />
particles/droplets. Upon requirement, TG and SE can be mobilized and serve as building<br />
blocks for membrane biosyn<strong>the</strong>sis. In a long-standing project <strong>of</strong> our laboratory, we investigate<br />
<strong>the</strong> characterization <strong>of</strong> enzymatic steps which lead to formation and mobilization <strong>of</strong> TG and<br />
SE depots.<br />
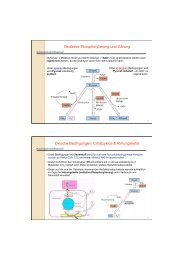
Life cycle <strong>of</strong> yeast neutral lipids: Genes involved in non-polar lipid formation and<br />
degradation.<br />
Previous studies in our laboratory had identified Tgl3p, Tgl4p and Tgl5p as major yeast TG<br />
lipases. In addition, however, a number <strong>of</strong> candidate gene products with potential<br />
lipase/esterase activity were also identified. One <strong>of</strong> <strong>the</strong>se polypeptides is Ldh1p, a lipid<br />
droplet associated hydrolytic enzyme. In collaboration with <strong>the</strong> laboratory <strong>of</strong> R. Erdmann,<br />
Bochum, Germany, we demonstrated that recombinant Ldh1p exerts esterase and TG lipase<br />
activities. The enzyme activity was abolished upon mutation <strong>of</strong> <strong>the</strong> conserved GXSXG-type<br />
lipase motif <strong>of</strong> <strong>the</strong> protein. Saccharomyces cerevisiae deleted <strong>of</strong> LDH1 was characterized by<br />
<strong>the</strong> appearance <strong>of</strong> giant lipid droplets and accumulation <strong>of</strong> non-polar lipids and phospholipids<br />
in LDs, indicative <strong>of</strong> a role <strong>of</strong> Ldh1p in maintaining lipid homeostasis.<br />
A major focus <strong>of</strong> our non-polar lipid project was <strong>the</strong> biochemical characterization <strong>of</strong> <strong>the</strong> three<br />
yeast TG lipases, Tgl3p, Tgl4p and Tgl5p. Previous work from our laboratory had<br />
demonstrated that deletion <strong>of</strong> TGL3 encoding <strong>the</strong> major yeast TG lipase resulted in decreased<br />
mobilization <strong>of</strong> TG, a sporulation defect and a changed pattern <strong>of</strong> fatty acids, especially<br />
increased amounts <strong>of</strong> C22:0 and C26:0 very long chain fatty acids in <strong>the</strong> TG fraction.<br />
Surprising evidence was obtained when <strong>the</strong> enzymology <strong>of</strong> <strong>the</strong> three TG lipases Tgl3p, Tgl4p<br />
18