Biennial Report 2016/2017
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
Selected Results<br />
High Energy Electron Irradiation of Gelatin Ferrogels for Bioapplications<br />
E. Wisotzki, A. Weidt, S. G. Mayr<br />
Ferrogels are emerging responsive materials that<br />
consist of a hydrogel matrix embedded with<br />
magnetic nanoparticles (MNP). Through coupling<br />
of the particles to the polymer network, ferrogels<br />
can deform, change rheology and alter swelling<br />
behavior in applied magnetic fields. With<br />
significant development in biocompatible<br />
hydrogels and MNP, ferrogels have gained<br />
attention for potential bioapplications such as soft<br />
actuators, drug delivery devices and tissue<br />
engineering scaffolds.<br />
In [1], high energy electron irradiation was<br />
demonstrated to crosslink gelatin hydrogels,<br />
resulting in thermal stability and a range of<br />
available mechanical properties, dependent on<br />
the irradiation dose. This reagent-free technique<br />
has the additional advantage of not requiring any<br />
(potentially toxic) chemical crosslinkers and has<br />
demonstrated low cytotoxicity in cell culture [2]. 10<br />
MeV electron beam irradiation was extended to<br />
gelatin ferrogels to investigate the ability to locally<br />
stabilize and confine such nanostructures into the<br />
gel network [2]. Using emerging methods of<br />
magnetic particle spectroscopy (MPS) and<br />
magnetorelaxometry (MRX) at the PTB Berlin, the<br />
thermal stability of these ferrogels was examined.<br />
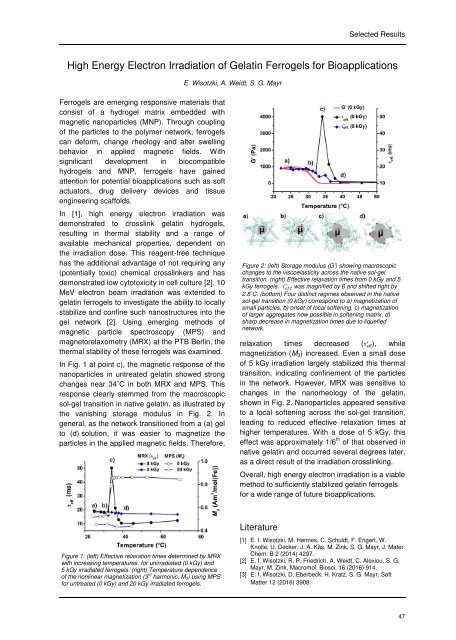
In Fig. 1 at point c), the magnetic response of the<br />
nanoparticles in untreated gelatin showed strong<br />
changes near 34˚C in both MRX and MPS. This<br />
response clearly stemmed from the macroscopic<br />
sol-gel transition in native gelatin, as illustrated by<br />
the vanishing storage modulus in Fig. 2. In<br />
general, as the network transitioned from a (a) gel<br />
to (d) solution, it was easier to magnetize the<br />
particles in the applied magnetic fields. Therefore,<br />
Figure 2: (left) Storage modulus (G’) showing macroscopic<br />
changes to the viscoelasticity across the native sol-gel<br />
transition. (right) Effective relaxation times from 0 kGy and 5<br />
Ȃ<br />
kGy ferrogels. was magnified by 6 and shifted right by<br />
2.8˚C. (bottom) Four distinct regimes observed in the native<br />
sol-gel transition (0 kGy) correspond to a) magnetization of<br />
small particles, b) onset of local softening, c) magnetization<br />
of larger aggregates now possible in softening matrix, d)<br />
sharp decrease in magnetization times due to liquefied<br />
network.<br />
relaxation times decreased Ȃ<br />
(̀ ), while<br />
magnetization (M 3 ) increased. Even a small dose<br />
of 5 kGy irradiation largely stabilized this thermal<br />
transition, indicating confinement of the particles<br />
in the network. However, MRX was sensitive to<br />
changes in the nanorheology of the gelatin,<br />
shown in Fig. 2. Nanoparticles appeared sensitive<br />
to a local softening across the sol-gel transition,<br />
leading to reduced effective relaxation times at<br />
higher temperatures. With a dose of 5 kGy, this<br />
effect was approximately 1/6 th of that observed in<br />
native gelatin and occurred several degrees later,<br />
as a direct result of the irradiation crosslinking.<br />
Overall, high energy electron irradiation is a viable<br />
method to sufficiently stabilized gelatin ferrogels<br />
for a wide range of future bioapplications.<br />
Figure 1: (left) Effective relaxation times determined by MRX<br />
with increasing temperatures, for unirradiated (0 kGy) and<br />
5 kGy irradiated ferrogels. (right) Temperature dependence<br />
of the nonlinear magnetization (3 rd harmonic, M 3) using MPS<br />
for untreated (0 kGy) and 20 kGy irradiated ferrogels.<br />
Literature<br />
[1] E. I. Wisotzki, M. Hennes, C. Schuldt, F. Engert, W.<br />
Knolle, U. Decker, J. A. Käs, M. Zink, S. G. Mayr, J. Mater.<br />
Chem. B 2 (2014) 4297.<br />
[2] E. I. Wisotzki, R. P. Friedrich, A. Weidt, C. Alexiou, S. G.<br />
Mayr, M. Zink, Macromol. Biosci. 16 (<strong>2016</strong>) 914.<br />
[3] E. I. Wisotzki, D. Eberbeck, H. Kratz, S. G. Mayr, Soft<br />
Matter 12 (<strong>2016</strong>) 3908.<br />
47