Jasmone 1 - ChemistforChrist
Jasmone 1 - ChemistforChrist
Jasmone 1 - ChemistforChrist
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
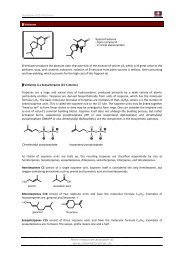
Synthesis of JASMONE<br />
Another way to see it is:<br />
Bn<br />
HO<br />
N<br />
S<br />
A nucleophilic Carbene<br />
This kind of carbenes are pretty important today:<br />
Ar The first carbene that was isolated, crystalized and characterized.<br />
N Today there are many chiral versions available that are used<br />
in asymmetric synthesis.<br />
N<br />
Ar<br />
N<br />
N<br />
Ar<br />
Ar<br />
Very reactive<br />
species! Prone<br />
to nucleophilic<br />
attack.<br />
N<br />
N<br />
Nu<br />
H<br />
N<br />
N<br />
N<br />
N<br />
Ar<br />
Ar<br />
6-π Electron aromatic<br />
system, that's why the carbene<br />
is so stable!<br />
Idea:<br />
Block the site prone<br />
to nucleophilic attack<br />
with very bulky substituents.<br />
The 2 plane where the 2 mesityl<br />
substituents are lying in are<br />
perpendicular to each other.<br />
More resources available at<br />
www.chemistforchrist.de<br />
Putting different charges on the<br />
same carbon atom can also be a<br />
way of representing a carbene!<br />
N<br />
N<br />
N<br />
N<br />
Mesityl- Adamantyl-<br />
The fact that carbenes can be used in catalysis as a Ligand is due to the fact that the 2 Electrons in the sp 2<br />
Orbital behave like lone pair and can thus donate electron density to the corresponding metal.<br />
R<br />
N<br />
N<br />
R<br />
behaves like a<br />
Donor e.g.<br />
R<br />
R P<br />
R<br />
Carbenes in catalysis are often named NHC-Ligands (N-Heterocyclic Carbenes) and offer many advantages<br />
compare to their Phosphorous counterparts:<br />
a) Environmentally more friendly (often non toxic)<br />
b) Not sensitive to oxidation<br />
c) Lower Molecular mass, for industrial purpose this means less gramms or kg of Ligand has to be added.