Jasmone 1 - ChemistforChrist
Jasmone 1 - ChemistforChrist
Jasmone 1 - ChemistforChrist
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
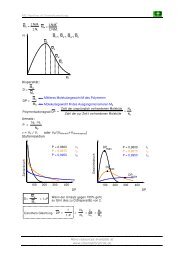
Synthesis of JASMONE<br />
The Nazarow reaction: Short revision<br />
For more details see: Org. React. 1994, 45, 1-158<br />
O<br />
O<br />
R R<br />
O<br />
H<br />
hν (254 nm)<br />
benzene<br />
AcOH, H 3PO 4<br />
50°C<br />
R R<br />
Disrotatory Conrotatory<br />
O<br />
R R<br />
R R<br />
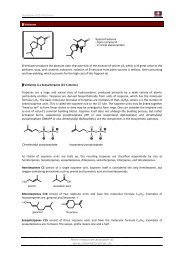
In its simplest form the Nazarow cyclization is the ring closure of a doubly α,β unsaturated ketone to give a<br />
cyclopentenone.<br />
O<br />
H<br />
R R<br />
O<br />
H<br />
R R<br />
R<br />
empty<br />
OH<br />
More resources available at<br />
www.chemistforchrist.de<br />
R<br />
O<br />
R R<br />
array of five p orbitals<br />
containing 4p electrons<br />
One of the five π orbitals involved is empty, so the cyclization is a 4π electrocyclic reaction and the orbitals<br />
forming the new σ bond must interact antarafacially.<br />
H<br />
O<br />
OH<br />
O<br />
OH<br />
OH<br />
R R<br />
R<br />
π 4<br />
a<br />
R<br />
H<br />
R<br />
R<br />
R R<br />
R R<br />
cyclopentenone<br />
Usually the double bond is formed at the site which is higher substituted (thermodynamically more stable<br />
product) to get the double bond at the less substituted site can also be done by introducing a siliconsubstituent:<br />
O<br />
TMS R<br />
Lewis acid<br />
LA<br />
O<br />
TMS R<br />
Nazarow<br />
LA<br />
O<br />
TMS R<br />
Anion<br />
H 3O<br />
O<br />
R