1 Selective serotonin reuptake inhibitors (SSRI) â sales, withdrawal ...
1 Selective serotonin reuptake inhibitors (SSRI) â sales, withdrawal ...
1 Selective serotonin reuptake inhibitors (SSRI) â sales, withdrawal ...
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
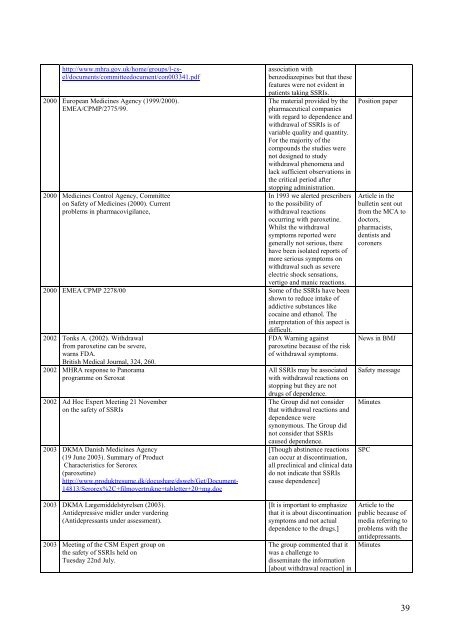
http://www.mhra.gov.uk/home/groups/l-csel/documents/committeedocument/con003341.pdf<br />
2000 European Medicines Agency (1999/2000).<br />
EMEA/CPMP/2775/99.<br />
2000 Medicines Control Agency, Committee<br />
on Safety of Medicines (2000). Current<br />
problems in pharmacovigilance,<br />
association with<br />
benzodiazepines but that these<br />
features were not evident in<br />
patients taking <strong>SSRI</strong>s.<br />
The material provided by the<br />
pharmaceutical companies<br />
with regard to dependence and<br />
<strong>withdrawal</strong> of <strong>SSRI</strong>s is of<br />
variable quality and quantity.<br />
For the majority of the<br />
compounds the studies were<br />
not designed to study<br />
<strong>withdrawal</strong> phenomena and<br />
lack sufficient observations in<br />
the critical period after<br />
stopping administration.<br />
In 1993 we alerted prescribers<br />
to the possibility of<br />
<strong>withdrawal</strong> reactions<br />
occurring with paroxetine.<br />
Whilst the <strong>withdrawal</strong><br />
symptoms reported were<br />
generally not serious, there<br />
have been isolated reports of<br />
more serious symptoms on<br />
<strong>withdrawal</strong> such as severe<br />
electric shock sensations,<br />
vertigo and manic reactions.<br />
2000 EMEA CPMP 2278/00 Some of the <strong>SSRI</strong>s have been<br />
shown to reduce intake of<br />
addictive substances like<br />
cocaine and ethanol. The<br />
interpretation of this aspect is<br />
difficult.<br />
2002 Tonks A. (2002). Withdrawal<br />
from paroxetine can be severe,<br />
warns FDA.<br />
British Medical Journal, 324, 260.<br />
2002 MHRA response to Panorama<br />
programme on Seroxat<br />
2002 Ad Hoc Expert Meeting 21 November<br />
on the safety of <strong>SSRI</strong>s<br />
2003 DKMA Danish Medicines Agency<br />
(19 June 2003). Summary of Product<br />
Characteristics for Serorex<br />
(paroxetine)<br />
http://www.produktresume.dk/docushare/dsweb/Get/Document-<br />
14813/Serorex%2C+filmovertrukne+tabletter+20+mg.doc<br />
FDA Warning against<br />
paroxetine because of the risk<br />
of <strong>withdrawal</strong> symptoms.<br />
All <strong>SSRI</strong>s may be associated<br />
with <strong>withdrawal</strong> reactions on<br />
stopping but they are not<br />
drugs of dependence.<br />
The Group did not consider<br />
that <strong>withdrawal</strong> reactions and<br />
dependence were<br />
synonymous. The Group did<br />
not consider that <strong>SSRI</strong>s<br />
caused dependence.<br />
[Though abstinence reactions<br />
can occur at discontinuation,<br />
all preclinical and clinical data<br />
do not indicate that <strong>SSRI</strong>s<br />
cause dependence]<br />
Position paper<br />
Article in the<br />
bulletin sent out<br />
from the MCA to<br />
doctors,<br />
pharmacists,<br />
dentists and<br />
coroners<br />
News in BMJ<br />
Safety message<br />
Minutes<br />
SPC<br />
2003 DKMA Lægemiddelstyrelsen (2003).<br />
Antidepressive midler under vurdering<br />
(Antidepressants under assessment).<br />
2003 Meeting of the CSM Expert group on<br />
the safety of <strong>SSRI</strong>s held on<br />
Tuesday 22nd July.<br />
[It is important to emphasize<br />
that it is about discontinuation<br />
symptoms and not actual<br />
dependence to the drugs.]<br />
The group commented that it<br />
was a challenge to<br />
disseminate the information<br />
[about <strong>withdrawal</strong> reaction] in<br />
Article to the<br />
public because of<br />
media referring to<br />
problems with the<br />
antidepressants.<br />
Minutes<br />
39