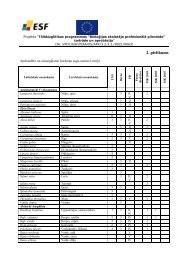
Fluorescence <strong>in</strong>tensity, a. u.Spektr<strong>of</strong>luorometrisko pētījumu metožu pielietojums smago metālu jonunoteikšanai ūdens vidēA.Pučk<strong>in</strong>s 1 , E.Kirilova 11 Daugavpils Universitāte, Daugavpils, Latvijae-mail: aleksandrs.puck<strong>in</strong>s@du.lvVides piesārņošana ar smagajiem metāliem mūsdienās ir ļoti aktuāla problēma. Tāpēcpašlaik ir pievērsta liela uzmanība, lai izstrādātu jaunas jutīgas šo vielu noteikšanasmetodes.Ņemot vērā z<strong>in</strong>āmo faktu, ka metālu joni var ietekmēt fluorescentā savienojuma,ar kuru saistās, fluorescences parametrus, tika veikts pētījums, kura laikā tika noskaidrotaspektr<strong>of</strong>luorometrisko metožu pielietojuma iespējamība smago metālu detektēšanai ūdensvidē.III600400200NiCoCu0400 500 600 700 800Wavelength, nmPētījumu laikā tika uzņemti vairāki krāsvielu fluorescences <strong>in</strong>tensitātes spektri, pēckuriem spriežot tika sec<strong>in</strong>āts, ka dažādu smago metālu klātbūtne var izraisīt dažādusefektus: gan organisko fluor<strong>of</strong>oru fluorescences pavāj<strong>in</strong>āšanu, gan pastipr<strong>in</strong>āšanu.Pētījuma gaitā tika noskaidrots, ka ar spektr<strong>of</strong>lorometriskas metodes palīdzību var veiktgan kvalitatīvo, gan kvantitatīvo dažu smago metālu noteikšanu ūdens vidē.Spektr<strong>of</strong>luorometrisko pētījumu metožu grupai ir raksturīga vesela virkne priekšrocību,kas nepiemīt pārējām analīzes metodēm un lietderīgi atšķir to no citām metodēm: zemasizmantošanas izmaksas, eksperimentu vienkāršums, metodes straujums un jūtīgums.- 14 -
Amid<strong>in</strong>es <strong>of</strong> Benzanthrone as Fluorescent Sensor for Metal IonsA.I.Puchk<strong>in</strong> 1 , I.D.Ivanova 2 , E.M.Kirilova 11 Chemistry Department, Daugavpils University, 13 Vienibas str., LV-5401, Daugavpils(Latvia);2 Faculty <strong>of</strong> Chemistry, University <strong>of</strong> Latvia, 48 Valdemara str., LV-1013, Riga (Latvia).e-mail: aleksandrs.puck<strong>in</strong>s@du.lvPollution <strong>of</strong> the environment by toxic metals is a major problem <strong>in</strong> the last decays due totheir <strong>in</strong>creas<strong>in</strong>g utilization <strong>in</strong> <strong>in</strong>dustry and agriculture.. These pollutants are discharged ortransported <strong>in</strong>to the atmosphere and aquatic and terrestrial environments and have thepotential to reach high concentrations. Large concentrations <strong>of</strong> some metal ions have beenshown to cause bra<strong>in</strong> damage, and are potentially lethal. Due to potential health risksassociated with exposure to metals, legal constra<strong>in</strong>ts on the allowed abundance <strong>of</strong> metalsare becom<strong>in</strong>g <strong>in</strong>creas<strong>in</strong>gly strict.As a result <strong>of</strong> the health threats posed by some metals, new methodologies that allow fortheir simple detection and quantitation are <strong>of</strong> <strong>in</strong>terest. Today many fluorescent <strong>organic</strong>sensors were designed for recognition and detection <strong>of</strong> heavy and transition metal ions.These chemosensors are compounds that <strong>in</strong>corporate a b<strong>in</strong>d<strong>in</strong>g site, a fluorophore, and ameans <strong>of</strong> communication between the two. The <strong>in</strong>teraction <strong>of</strong> a metal ion with an <strong>organic</strong>ligand may result <strong>in</strong> fluorescent enhancement or fluorescence quench<strong>in</strong>g.Our recent efforts have been focused largely on synthesiz<strong>in</strong>g novel <strong>organic</strong> fluorophoressuch as N-conta<strong>in</strong><strong>in</strong>g benzanthrone derivatives [1,2]. The present work focuses on thespectral characterization <strong>of</strong> benzanthronylamid<strong>in</strong>es and its complexes with transitionmetal ions. Obta<strong>in</strong>ed results testify that <strong>in</strong>vestigated dyes can be used as sensitivefluorescent chemosensors for transition metals: the one <strong>of</strong> them displays positive response(3-7-fold fluorescence <strong>in</strong>creas<strong>in</strong>g) to the presence <strong>of</strong> Ni 2+ or Co 2+ ions, but somesynthesized amid<strong>in</strong>es show negative response (quench<strong>in</strong>g) <strong>in</strong> fluorescence <strong>in</strong>tensity to Crions. Further <strong>in</strong>vestigations can lead to development <strong>of</strong> new method for the detection <strong>of</strong>some cations.1. E.M Kirilova, S. V Belyakov, I. Kaln<strong>in</strong>a. <strong>in</strong>: G. Vayssilov, R. Nikolova (Eds), Topics <strong>in</strong>Chemistry & Materials Science, S<strong>of</strong>ia: Heron Press, 3, (2009) 19-28.2. V.M. Trusova, E. Kirilova, I. Kaln<strong>in</strong>a, G. Kirilov, O.A. Zhytniakivska, P.V.Fedorov, G.Gorbenko. J.Fluoresc. 22 (2012) 953-959.- 15 -