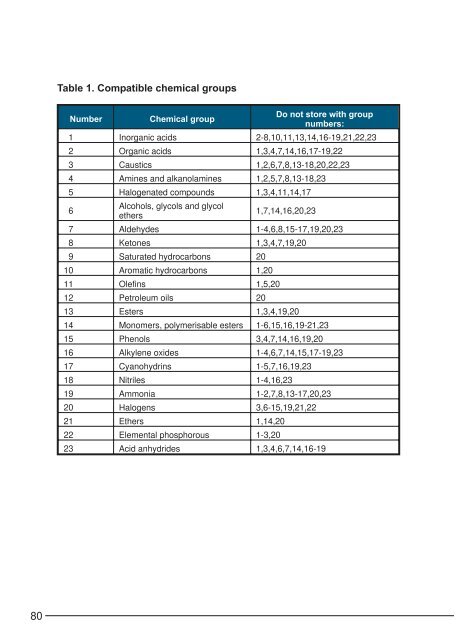
Table 1. Compatible chemical groupsNumberChemical groupDo not s<strong>to</strong>re with groupnumbers:1 In<strong>org</strong>anic acids 2-8,10,11,13,14,16-19,21,22,232 Organic acids 1,3,4,7,14,16,17-19,223 Caustics 1,2,6,7,8,13-18,20,22,234 Am<strong>in</strong>es and alkanolam<strong>in</strong>es 1,2,5,7,8,13-18,235 Halogenated compounds 1,3,4,11,14,176Alcohols, glycols and glycole<strong>the</strong>rs1,7,14,16,20,237 Aldehydes 1-4,6,8,15-17,19,20,238 Ke<strong>to</strong>nes 1,3,4,7,19,209 Saturated hydrocarbons 2010 Aromatic hydrocarbons 1,2011 Olef<strong>in</strong>s 1,5,2012 Petroleum oils 2013 Esters 1,3,4,19,2014 Monomers, polymerisable esters 1-6,15,16,19-21,2315 Phenols 3,4,7,14,16,19,2016 Alkylene oxides 1-4,6,7,14,15,17-19,2317 Cyanohydr<strong>in</strong>s 1-5,7,16,19,2318 Nitriles 1-4,16,2319 Ammonia 1-2,7,8,13-17,20,2320 Halogens 3,6-15,19,21,2221 E<strong>the</strong>rs 1,14,2022 Elemental phosphorous 1-3,2023 Acid anhydrides 1,3,4,6,7,14,16-1980
Table 2.Examples <strong>of</strong> <strong>in</strong>compatible comb<strong>in</strong>ations <strong>of</strong> some commonlyused chemicalsChemicalAscetic acidAcetyleneAlkali metals (e.g.sodiumAmmonia,anhydrousAmmonium nitrateAnil<strong>in</strong>eBrom<strong>in</strong>eCarbon, activatedChloratesChromic acidChlor<strong>in</strong>eCopperFlammable liquidsHydrocarbonsHydr<strong>of</strong>louric acidHydrogen peroxideHydrogen sulphideIod<strong>in</strong>eMercuryNitric acidOxalic acidPerchloric acidEnsure no contact withchromic acid, nitric acid, hydroxyl compounds, perchloric acid,peroxides, permanganatechlor<strong>in</strong>e, brom<strong>in</strong>e, copper, fluor<strong>in</strong>e, silver, mercurywater, chlor<strong>in</strong>ated hydrocarbons, carbon dioxide, halogensmercury, chlor<strong>in</strong>e, calcium hypochlorite, iod<strong>in</strong>e, brom<strong>in</strong>e,hydr<strong>of</strong>luoric acidacids, metal powders, flammable liquids, chlorates, nitrites,sulphur, f<strong>in</strong>ely divided combustible materialsnitric acid, hydrogen peroxidesame as chlor<strong>in</strong>ecalcium hypochlorite, all oxidis<strong>in</strong>g agentsammonium salts, acids, metal powders, sulphur, f<strong>in</strong>ely dividedcombustible materialsascetic acid, naphthalene, camphor, glycer<strong>in</strong>, turpent<strong>in</strong>e,alcohol, flammable liquidsammonia, acetylene, butadiene, butane, methane, propane (oro<strong>the</strong>r petroleum gases), hydrogen, sodium carbide, turpent<strong>in</strong>e,benzene, f<strong>in</strong>ely divided metalsacetylene, hydrogen peroxideammonium nitrate, <strong>in</strong><strong>org</strong>anic acids, hydrogen peroxide, sodiumperoxide, halogensfluor<strong>in</strong>e, chlor<strong>in</strong>e, brom<strong>in</strong>e, chromic acid, sodium peroxideanhydrous ammonia, ammonium hydroxidecopper, chromium, iron, most metals or <strong>the</strong>ir salts, alcohols,ace<strong>to</strong>ne, anil<strong>in</strong>e, nitromethane, flammable liquids, oxidis<strong>in</strong>ggasesfum<strong>in</strong>g nitric acid, oxidis<strong>in</strong>g gasesacetylene, ammonia (aqueous or anhydrous), hydrogenacetylene, fulm<strong>in</strong>ic acid, ammoniaacetic acid, anil<strong>in</strong>e, chromic acid, hydrocyanic acid, hydrogensulphide, flammable liquids, flammable gasessilver, mercuryacetic anhydride, bismuth and its alloys, <strong>org</strong>anic materials81