8. H.K. Das, <strong>Eng<strong>in</strong>eer<strong>in</strong>g</strong> Mathematics.9. Vijai Shankar Verma & Sanjeev Kumar, <strong>Eng<strong>in</strong>eer<strong>in</strong>g</strong> Mathematics.10. Rakesh Dubey, <strong>Eng<strong>in</strong>eer<strong>in</strong>g</strong> MathematicsCHEMISTRYCourse Code CHEM-513 CREDIT : 5 (3-1-1)1. General Chemistry:Advanced Theory of Chemical Bond<strong>in</strong>g: Valence bond and molecularorbital theory. Structure of NH 3 , H 2 O, SO 3 , PCl 5 , XeO 2 molecules. Theories of bond<strong>in</strong>g <strong>in</strong>metals and semiconductors,n-type and p-type semi-conductors, Imperfections <strong>in</strong> materials.Born-Haber cycle, Bragg’s conditions.2. Physical Chemistry:Equilibrium on Reactivity: Bronsted and Lewis Acids, pH, pka, pkbScale, Buffer solution. Stereochemistry of organic compounds, Co-ord<strong>in</strong>ation chemistry,Nomenclature, Valence Bond and crystal field theory.Chemical K<strong>in</strong>etics & Catalysis: Rate law, Order of reactions, Parallel and reversiblereactions, Catalysis, Homogeneous and heterogeneous catalysis, Characteristics ofcatalytic reaction, Catalytic promoters and poi-sons, Auto catalysis and negativecatalysis, Intermediate compound formation theory and absorption theory.3. Environment Chemistry:Atmospheric Chemistry & Air Pollution: Environment andEcology, Environmental segments, Structure and composition of atmosphere, RadiationBalance of Earth and Green House Effect, Formation and depletion of Ozone layer,Chemical and photochemical reactions of various species <strong>in</strong> atmosphere, Air pollution –sources, reactions and s<strong>in</strong>ks for pollutants, Acid ra<strong>in</strong>s and Smog formation. Pollutioncontrol methods.Corrosion and Lubrication: Introduction, causes of corrosion, Theories of corrosion,Factors <strong>in</strong>fluenc<strong>in</strong>g Corrosion, Corrosion <strong>in</strong>hibitors, passivity, Types of corrosion,Protection from corrosion and protective coat<strong>in</strong>gs. Theory, Classification and mechanismof Lubrication.4. Applied Chemistry:Water and Waste Water Chemistry: Introduction, Hardness ofwater, characteristics imparted by impurities, Analysis of contam<strong>in</strong>ants, Treatment ofWater by Zeolite, L-S process, Boiler feed water, Waste water treatment.5. Chemistry of <strong>Eng<strong>in</strong>eer<strong>in</strong>g</strong> Materials:Fuels & Combustion: Classification of fuels, Nonconventional Energy, Biogas, Biomass and solar energy. Calorific value- gross and net,characteristics of good fuel, Determ<strong>in</strong>ation of calorific value, Solid fuels, Analysis of coal,Liquid fuels.Instrumentation: IR, UV, NMR, MASS AND ASS.6. <strong>Industrial</strong> Chemistry:Polymer Chemistry: Classification of Polymers, Includ<strong>in</strong>gBiopolymers condensation and addition polymers and their applications. <strong>Industrial</strong>Application and mechanism of chemical reaction, Beckman, Hoffman, Reimer Tiemann,Cunnizzaro, Diels Alder and Skraup synthesis.References:1. Puri and Sharma/Pr<strong>in</strong>ciples of Physical Chemistry.2. Manas Chandra/Atomic Structure and Chemical Bond.3. Bahl and Tuli /<strong>Eng<strong>in</strong>eer<strong>in</strong>g</strong> Chemistry.4. Ja<strong>in</strong> and Ja<strong>in</strong>/A Text-Book of <strong>Eng<strong>in</strong>eer<strong>in</strong>g</strong> Chemistry5. S.S Dara/Environmental Chemistry and Pollution Control.6. S.S Dara /Environmental Chemistry.7. A.K De/Environmental Chemistry.LIST OF EXPERIMENTS (ANY TEN):1. To determ<strong>in</strong>e the percentage of available chlor<strong>in</strong>e <strong>in</strong> the supplied sample of Bleach<strong>in</strong>gpowder.
2. To determ<strong>in</strong>e the Ferrous content <strong>in</strong> the supplied sample of iron ore by titrimetric analysisaga<strong>in</strong>st standard K Cr solution us<strong>in</strong>g K, Fe(CN) g as external <strong>in</strong>dicator.3. To determ<strong>in</strong>e the chloride content <strong>in</strong> supplied water sample us<strong>in</strong>g Mohr’s method.4. To determ<strong>in</strong>e the constituents and amount of alkal<strong>in</strong>ity of the supplied water sample.5. To determ<strong>in</strong>e the Temporary and Permanent hardness of water sample by Coplexometry.6. To f<strong>in</strong>d the Chemical Oxygen Demand of a waste water sample us<strong>in</strong>g Potassium dichromate.7. To determ<strong>in</strong>e iron concentration <strong>in</strong> the sample of water by spectrophotometric method.8. To f<strong>in</strong>d out the Velocity constant for the <strong>in</strong>version of cane sugar <strong>in</strong> acidic medium and toshow that <strong>in</strong>version follows the first order k<strong>in</strong>etics.9. To determ<strong>in</strong>e the Molecular weight of a polystyrene sample by us<strong>in</strong>g Viscometer method.10. To determ<strong>in</strong>e pH of a solution us<strong>in</strong>g a pH-meter and titration of such a solution pHmetrically.11. To determ<strong>in</strong>e the calorific value of a fuel sample by us<strong>in</strong>g a Bomb Calorimeter.12. Analysis of a coal sample by proximate analysis method.References:1. Vogel’s Qualitative Chemical Analysis: Ed. By Jaffery Bassette et. al. (ELBS).2. Applied Chemistry- Theory and Practice, 2 nd Ed. By Virmani and Narula (New AgeInternational Pub.).3. Experiments <strong>in</strong> <strong>Eng<strong>in</strong>eer<strong>in</strong>g</strong> Chemistry, Ed. By Masood Alam (Maktaba Jamia Limited).BASIC ELECTRONICS1-1)Course Code ECE-301 CREDIT : 4 (2-1. Energy Bands <strong>in</strong> Solids: Energy band theory of solids, Concept of forbidden gap, Insulators,Metals and Semiconductors.2. Transport Phenomenon <strong>in</strong> Semiconductors: Mobility and conductivity, electrons and holes <strong>in</strong>an <strong>in</strong>tr<strong>in</strong>sic semiconductor, Donor and acceptor impurities, Fermi level, carrier densities <strong>in</strong>semiconductor, electrical properties of semiconductor, Hall effect, Diffusion.3. Junction Diode:P-N junction, depletion layer, V-I characteristics, diode resistance,capacitance, switch<strong>in</strong>g time, diode application as a rectifier (half wave and full wave), diodecircuits (clipper, clamper, voltage multipliers) Breakdown mechanism, Zener & Avalanche,breakdown characteristics, Zener diode and its applications.4. Bi-junction Transistor:Bipolar junction Transistor, CE, CB and CC configuration,characteristic curves (cut off, active and saturation region), Requirement of bias<strong>in</strong>g, bias<strong>in</strong>g typesand bias<strong>in</strong>g analysis, stability.5. Transistor as an Amplifier:Graphical analysis of CE amplifier, concept of voltage ga<strong>in</strong>,current ga<strong>in</strong> and power ga<strong>in</strong>, h-parameter (low frequency), computation of A v , R I , R o andapproximate formulae.6. Operational Amplifiers:Concepts of ideal op-amp, <strong>in</strong>vert<strong>in</strong>g, non-<strong>in</strong>vert<strong>in</strong>g and unity ga<strong>in</strong>amplifiers, adders, difference amplifiers. , Integrators.
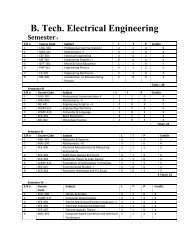
- Page 1 and 2: B. Tech. Production & IndustrialEng
- Page 3 and 4: 6 ME 516 Energy Management 3‐1‐
- Page 5 and 6: Sin, orign of sin, manifestation of
- Page 7: Center of gravity, centroids of lin
- Page 11 and 12: 6. Study of Scientific and General
- Page 13 and 14: 6. X-Rays: Origin of X-rays, Contin
- Page 15 and 16: 11. Write C program to demonstrate
- Page 17 and 18: Steam Turbines: Classification, imp
- Page 19 and 20: Theories of Failure:Various theorie
- Page 21 and 22: Environment Protection act.Air (Pre
- Page 23 and 24: Transformers:Construction, EMF equa
- Page 25 and 26: 5. Production Planning and Control:
- Page 27 and 28: Interpolation:Introduction, Errors
- Page 29 and 30: Note-1. Students may be advised to
- Page 31 and 32: Mental blocks, Removal blocks, Idea
- Page 33 and 34: Carburetion and gasoline Injection:
- Page 35 and 36: With initial review teaching of Aut
- Page 37 and 38: 2. Conventional energy resources an
- Page 39 and 40: Array, matrix, string, structure, c
- Page 41 and 42: Generals requirements, Road, tyre a
- Page 43 and 44: INTRODUCTION : Past , Present and f
- Page 45 and 46: Study of the design aspects. Fluid
- Page 47: MECHANICAL VIBRATIONCourse Code ME-