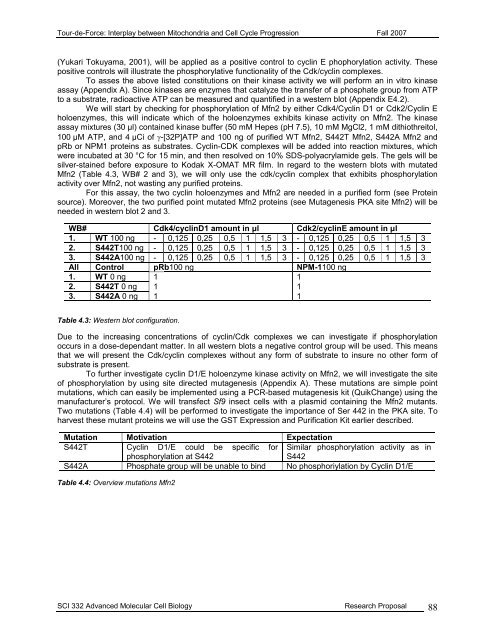
<strong>Tour</strong>-<strong>de</strong>-<strong>Force</strong>: Interplay between Mitochondria and Cell Cycle Progression Fall 2007(Yukari Tokuyama, 2001), will be applied as a positive control to cyclin E phophorylation activity. Thesepositive controls will illustrate the phosphorylative functionality of the Cdk/cyclin complexes.To asses the above listed constitutions on their kinase activity we will perform an in vitro kinaseassay (Appendix A). Since kinases are enzymes that catalyze the transfer of a phosphate group from ATPto a substrate, radioactive ATP can be measured and quantified in a western blot (Appendix E4.2).We will start by checking for phosphorylation of Mfn2 by either Cdk4/Cyclin D1 or Cdk2/Cyclin Eholoenzymes, this will indicate which of the holoenzymes exhibits kinase activity on Mfn2. The kinaseassay mixtures (30 µl) contained kinase buffer (50 mM Hepes (pH 7.5), 10 mM MgCl2, 1 mM dithiothreitol,100 µM ATP, and 4 µCi of γ-[32P]ATP and 100 ng of purified WT Mfn2, S442T Mfn2, S442A Mfn2 andpRb or NPM1 proteins as substrates. Cyclin-CDK complexes will be ad<strong>de</strong>d into reaction mixtures, whichwere incubated at 30 °C for 15 min, and then resolved on 10% SDS-polyacrylami<strong>de</strong> gels. The gels will besilver-stained before exposure to Kodak X-OMAT MR film. In regard to the western blots with mutatedMfn2 (Table 4.3, WB# 2 and 3), we will only use the cdk/cyclin complex that exhibits phosphorylationactivity over Mfn2, not wasting any purified proteins.For this assay, the two cyclin holoenzymes and Mfn2 are nee<strong>de</strong>d in a purified form (see Proteinsource). Moreover, the two purified point mutated Mfn2 proteins (see Mutagenesis PKA site Mfn2) will benee<strong>de</strong>d in western blot 2 and 3.WB# Cdk4/cyclinD1 amount in µl Cdk2/cyclinE amount in µl1. WT 100 ng - 0,125 0,25 0,5 1 1,5 3 - 0,125 0,25 0,5 1 1,5 32. S442T100 ng - 0,125 0,25 0,5 1 1,5 3 - 0,125 0,25 0,5 1 1,5 33. S442A100 ng - 0,125 0,25 0,5 1 1,5 3 - 0,125 0,25 0,5 1 1,5 3All Control pRb100 ng NPM-1100 ng1. WT 0 ng 1 12. S442T 0 ng 1 13. S442A 0 ng 1 1Table 4.3: Western blot configuration.Due to the increasing concentrations of cyclin/Cdk complexes we can investigate if phosphorylationoccurs in a dose-<strong>de</strong>pendant matter. In all western blots a negative control group will be used. This meansthat we will present the Cdk/cyclin complexes without any form of substrate to insure no other form ofsubstrate is present.To further investigate cyclin D1/E holoenzyme kinase activity on Mfn2, we will investigate the siteof phosphorylation by using site directed mutagenesis (Appendix A). These mutations are simple pointmutations, which can easily be implemented using a PCR-based mutagenesis kit (QuikChange) using themanufacturer’s protocol. We will transfect Sf9 insect cells with a plasmid containing the Mfn2 mutants.Two mutations (Table 4.4) will be performed to investigate the importance of Ser 442 in the PKA site. Toharvest these mutant proteins we will use the GST Expression and Purification Kit earlier <strong>de</strong>scribed.Mutation Motivation ExpectationS442T Cyclin D1/E could be specific for Similar phosphorylation activity as inphosphorylation at S442S442S442A Phosphate group will be unable to bind No phosphoriylation by Cyclin D1/ETable 4.4: Overview mutations Mfn2SCI 332 Advanced Molecular Cell Biology Research Proposal 88
<strong>Tour</strong>-<strong>de</strong>-<strong>Force</strong>: Interplay between Mitochondria and Cell Cycle Progression Fall 2007DiscussionBy performing the proposed research, the un<strong>de</strong>rstanding of several basic cell biological issues, such ascell cycle arrest, cell cycle progression and oxidative phosphorylation will be enriched. Through Mfn2 alink might be found between cell cycle progression and mitochondrial activity. However, this link to the cellcycle is not the only reason the proposed research is relevant. Mfn2 is mutated in variousneuro<strong>de</strong>generative diseases, of which Charcot-Marie-Tooth disease is the most prevalently studied.Moreover, since <strong>de</strong>creased levels of cytosolic Mfn2 have been noticed in hyper-proliferative diseases,insight into this protein might help to <strong>de</strong>velop new medical therapies.However, research is a dynamic process, and thus the proposed research does not provi<strong>de</strong> a completepicture. Even if all our hypotheses are indicated to be true, further research has to be done. The secondhypothesis will gain insight into the function of the Mfn2-Stoml2 complex. It could, however, still beexplored what the binding site of Stoml2 to Mfn2 is. In addition, this research doesn’t go into the exactpathway through which Mfn2 is able to regulate OXPHOS. Further research could investigate throughwhat mechanism this occurs.Hypothesis 3 will provi<strong>de</strong> insight into the formation of cytosolic Mfn2. It is suggested to conductadditional experiments that explore the conditions un<strong>de</strong>r which mitochondrial Mfn2 is cleaved to formcytosolic Mfn2. As proposed in the introduction, when cell cycle arrest is nee<strong>de</strong>d, there might be amechanism that cleaves the mitochondrial isoform of Mfn2. This will result in a <strong>de</strong>crease of mitochondrialMfn2, as well as an increase in cytosolic Mfn2 levels. Therefore, a bi-functional mechanism will work toachieve cell cycle arrest or apoptosis. Although this research will point out whether cleavage occurs, moreresearch will have to be done to <strong>de</strong>termine whether this proposed mechanism is true. It could beinvestigated where cleavage occurs, as this might occur either in the cytosol or after insertion into themitochondrial membrane. Furthermore, it can be investigated which protein(s) are involved in this processof cleavage. Additionally, it can be explored un<strong>de</strong>r which cellular conditions cleavage is upregulated.The fourth hypothesis focuses on the relation between Ras, cytosolic Mfn2 and cell cycle arrest.This research will show un<strong>de</strong>r what conditions the binding of Mfn2 to Ras causes cell cycle arrest,however it doesn’t go into the binding mechanisms. Further research could investigate this.If hypothesis 5 proves to be true, thus, if there is a correlation between mitochondrial or cytosolicMfn2 levels and cyclin D1 and cyclin E, further research is nee<strong>de</strong>d to see whether this correlationindicates causality. As a follow up experiment, the first step would be to look at the activity of the cyclins,since this <strong>de</strong>pends on their interaction with cdk’s. At the indicated time intervals co-immunoprecipitation ofthe cyclins and their cdk’s (cdk4 and cdk2, respectively) is suggested in or<strong>de</strong>r to extract the activecomplexes. An in vitro kinase assay would thereafter be necessary to quantify the complexes and tocheck whether the cyclins are in<strong>de</strong>ed more active. We would have liked to inclu<strong>de</strong> these experiments inour research, however due to time limitations this is beyond the scope of our project.However, this hypothesis might show that cyclin D1 / E directly influence Mfn2 by phosphorylation.Whether this phosphorylation results in activation or <strong>de</strong>activation has to be explored. In case of<strong>de</strong>activation, this could mean that Mfn-2 is no longer able to bind to Ras and subsequently induce cellcycle arrest. In addition, the protein would no longer be able to mediate OXPHOS. Both would be<strong>de</strong>sirable in progression from G1 to S phase. Since experiment 5.2 will be conducted in vitro, the resultscannot be generalized to in vivo circumstances. Further research could explore the relation in vivo.SCI 332 Advanced Molecular Cell Biology Research Proposal 89