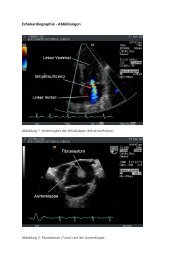
Jahresbericht der Klinik - Klinik für Kardiologie - UniversitätsSpital ...
Jahresbericht der Klinik - Klinik für Kardiologie - UniversitätsSpital ...
Jahresbericht der Klinik - Klinik für Kardiologie - UniversitätsSpital ...
Sie wollen auch ein ePaper? Erhöhen Sie die Reichweite Ihrer Titel.
YUMPU macht aus Druck-PDFs automatisch weboptimierte ePaper, die Google liebt.
Francesco Cosentino, MD, PhD<br />
Research Group Lea<strong>der</strong> & Visiting Prof.<br />
Basic Cardiovascular Research<br />
Diabetes Mellitus and the Vessel Wall<br />
Patients with diabetes are at risk of cardiovascular diseases (CVD). It<br />
was reported that p66 Shc protein is involved in modulation of oxidative<br />
stress. Mice lacking p66 Shc (p66 Shc−/− ) have increased resistance<br />
to reactive oxygen species (ROS) and prolonged life span. Although<br />
the exact biochemical role of p66 Shc has yet to be determined, it participates<br />
in mitochondrial ROS production. In response to stress signals,<br />
p66 Shc is activated by PKC and enters the mitochondrion, where<br />
it leads to accumulation of ROS and cellular death. We demonstrated<br />
that p66 Shc activation is crucial for induction of diabetic vascular dysfunction.<br />
Indeed, p66 Shc deletion prevents endothelial dysfunction<br />
and oxidative stress in diabetic mice. p66 Shc−/− diabetic mice showed<br />
lower ROS which accounted for the preserved NO release.<br />
Recently, we focused on the molecular mechanisms un<strong>der</strong>lying the<br />
long-lasting effects of hyperglycemia despite optimal glucose control.<br />
This investigation relies on clinical trials showing that intensive<br />
glycemic control has failed to reduce the burden of cardiovascular<br />
disease in diabetes. Of note, glucose-lowering therapy was started<br />
after a duration of diabetes ranging from 8 to 11 years. By contrast,<br />
early treatment of hyperglycemia reduces myocardial infarction and<br />
mortality. These observations support the hypothesis that hyperglycemic<br />
environment is remembered in the vasculature. ROS are likely<br />
involved in this «memory» phenomenon but the signaling pathways<br />
responsible for persistence of ROS production after glucose normalization<br />
are unknown.<br />
Hyperglycemia<br />
Endothelial cell<br />
Hyperglycemic<br />
Memory<br />
Normoglycemia<br />
PKCβ₂<br />
p66Shc NADPH<br />
ROS<br />
Since human and experimental diabetes have been associated with<br />
upregulation of p66 Shc , we hypothesize that its activation may continue<br />
despite glucose normalization, perpetuating redox vascular<br />
damage. Our preliminary data show that restoration of normal glucose<br />
levels does not affect p66 Shc upregulation, or ROS increase in<br />
human endothelial cells [Fig. 1]. Furthermore, we targeted p66 Shc at<br />
the time of glucose normalization. Its selective knockdown resulted<br />
in less oxidative stress and apoptosis, suggesting p66 Shc plays a role<br />
in hyperglycemic memory.<br />
NO<br />
Endothelial Dysfunction<br />
Vascular Damage<br />
Cardiovascular Events<br />
Fig. 1 Schematic representation of p66Shc critical involvement<br />
in vascular hyperglycemic memory<br />
We have confirmed these findings in vivo showing that restoration of<br />
normoglycemia by insulin does not revert p66 Shc activation in diabetic<br />
mice. Our investigation will elucidate upstream signaling pathways<br />
linking persistent hyperglycemic stress to ROS generation and, hence,<br />
CVD. Thus, a better un<strong>der</strong>standing of p66 Shc appears to be of utmost<br />
importance and may provide a new therapeutic avenue to reduce<br />
cardiovascular health burden.<br />
Fig. 2 Francesco Cosentino (MD, PhD) and his research group<br />
(left to right): Sarah Costantino (PhD student), Francesco<br />
Cosentino (MD, PhD), Francesco Paneni (MD, PhD student)<br />
and Elena Osto (MD, PhD)