Spectroscopy in a Suitcase - Royal Society of Chemistry
Spectroscopy in a Suitcase - Royal Society of Chemistry
Spectroscopy in a Suitcase - Royal Society of Chemistry
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
MASS SPECTROMETRY (MS)<br />
INTRODUCTION 3<br />
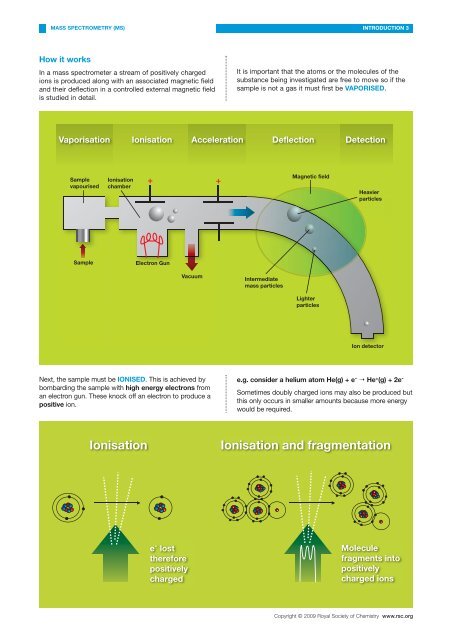
How it works<br />
In a mass spectrometer a stream <strong>of</strong> positively charged<br />
ions is produced along with an associated magnetic field<br />
and their deflection <strong>in</strong> a controlled external magnetic field<br />
is studied <strong>in</strong> detail.<br />
It is important that the atoms or the molecules <strong>of</strong> the<br />
substance be<strong>in</strong>g <strong>in</strong>vestigated are free to move so if the<br />
sample is not a gas it must first be VAPORISED.<br />
Vaporisation Ionisation Acceleration Deflection Detection<br />
Sample<br />
vapourised<br />
Ionisation<br />
chamber<br />
+ +<br />
Magnetic field<br />
Heavier<br />
particles<br />
Sample<br />
Electron Gun<br />
Vacuum<br />
Intermediate<br />
mass particles<br />
Lighter<br />
particles<br />
Ion detector<br />
Next, the sample must be IONISED. This is achieved by<br />
bombard<strong>in</strong>g the sample with high energy electrons from<br />
an electron gun. These knock <strong>of</strong>f an electron to produce a<br />
positive ion.<br />
e.g. consider a helium atom He(g) + e - He + (g) + 2e -<br />
Sometimes doubly charged ions may also be produced but<br />
this only occurs <strong>in</strong> smaller amounts because more energy<br />
would be required.<br />
Ionisation<br />
Ionisation and fragmentation<br />
e - lost<br />
therefore<br />
positively<br />
charged<br />
Molecule<br />
fragments <strong>in</strong>to<br />
positively<br />
charged ions<br />
Copyright © 2009 <strong>Royal</strong> <strong>Society</strong> <strong>of</strong> <strong>Chemistry</strong> www.rsc.org