Radar Technology for Level Gauging - Krohne
Radar Technology for Level Gauging - Krohne
Radar Technology for Level Gauging - Krohne
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
● Cyclic compounds because of their high symmetry of faces have lower εr values.<br />
3. <strong>Radar</strong>-Füllstandsmesssysteme<br />
● The dielectric characteristic of nitrogen or halogen derivatives varies considerably,<br />
and depends essentially on the symmetry in the molecule. Dichlorobenzene is a case<br />
in point, with the 3 positional isomers having ε r values of between 2.5 and 10.<br />
A table with ε r values of different products can be found in Annex A.<br />
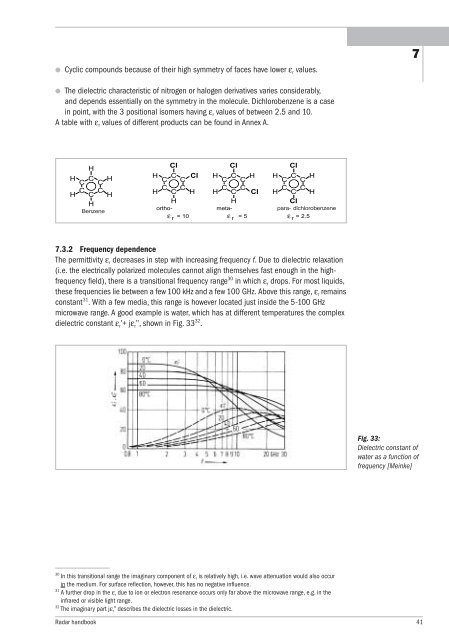
7.3.2 Frequency dependence<br />
The permittivity ε r decreases in step with increasing frequency f. Due to dielectric relaxation<br />
(i.e. the electrically polarized molecules cannot align themselves fast enough in the highfrequency<br />
field), there is a transitional frequency range 30 in which ε r drops. For most liquids,<br />
these frequencies lie between a few 100 kHz and a few 100 GHz. Above this range, ε r remains<br />
constant 31 .With a few media, this range is however located just inside the 5-100 GHz<br />
microwave range. A good example is water, which has at different temperatures the complex<br />
dielectric constant ε r’+ jε r’’, shown in Fig. 33 32 .<br />
30 In this transitional range the imaginary component of εr is relatively high, i.e. wave attenuation would also occur<br />
in the medium. For surface reflection, however, this has no negative influence.<br />
31 A further drop in the εr due to ion or electron resonance occurs only far above the microwave range, e.g. in the<br />
infrared or visible light range.<br />
32 The imaginary part jεr” describes the dielectric losses in the dielectric.<br />
<strong>Radar</strong> handbook 41<br />
7<br />
Fig. 33:<br />
Dielectric constant of<br />
water as a function of<br />
frequency [Meinke]