Monograph - Metrohm
Monograph - Metrohm
Monograph - Metrohm
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
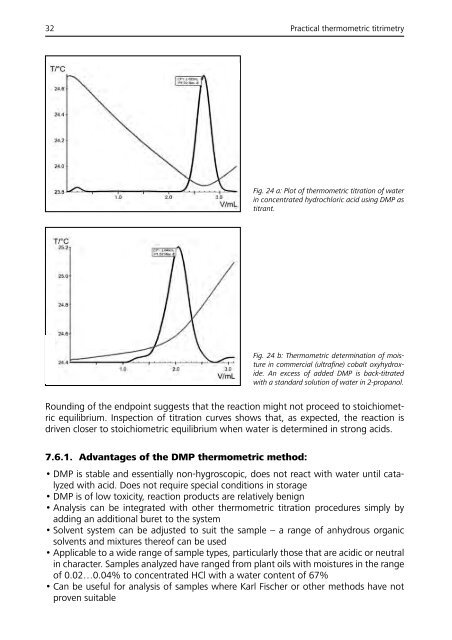
32 Practical thermometric titrimetry<br />
Fig. 24 a: Plot of thermometric titration of water<br />
in concentrated hydrochloric acid using DMP as<br />
titrant.<br />
Fig. 24 b: Thermometric determination of moisture<br />
in commercial (ultrafine) cobalt oxyhydroxide.<br />
An excess of added DMP is back-titrated<br />
with a standard solution of water in 2-propanol.<br />
Rounding of the endpoint suggests that the reaction might not proceed to stoichiometric<br />
equilibrium. Inspection of titration curves shows that, as expected, the reaction is<br />
driven closer to stoichiometric equilibrium when water is determined in strong acids.<br />
7.6.1. Advantages of the DMP thermometric method:<br />
• DMP is stable and essentially non-hygroscopic, does not react with water until catalyzed<br />
with acid. Does not require special conditions in storage<br />
• DMP is of low toxicity, reaction products are relatively benign<br />
• Analysis can be integrated with other thermometric titration procedures simply by<br />
adding an additional buret to the system<br />
• Solvent system can be adjusted to suit the sample – a range of anhydrous organic<br />
solvents and mixtures thereof can be used<br />
• Applicable to a wide range of sample types, particularly those that are acidic or neutral<br />
in character. Samples analyzed have ranged from plant oils with moistures in the range<br />
of 0.02…0.04% to concentrated HCl with a water content of 67%<br />
• Can be useful for analysis of samples where Karl Fischer or other methods have not<br />
proven suitable