Prescribed Drugs Provider Manual - Iowa Department of Human ...
Prescribed Drugs Provider Manual - Iowa Department of Human ...
Prescribed Drugs Provider Manual - Iowa Department of Human ...
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
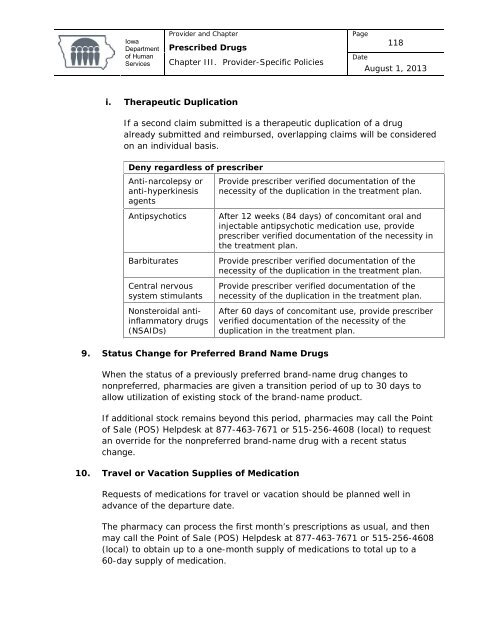
<strong>Iowa</strong><strong>Department</strong><strong>of</strong> <strong>Human</strong>Services<strong>Provider</strong> and Chapter<strong>Prescribed</strong> <strong>Drugs</strong>Chapter III. <strong>Provider</strong>-Specific PoliciesPage118DateAugust 1, 2013i. Therapeutic DuplicationIf a second claim submitted is a therapeutic duplication <strong>of</strong> a drugalready submitted and reimbursed, overlapping claims will be consideredon an individual basis.Deny regardless <strong>of</strong> prescriberAnti-narcolepsy oranti-hyperkinesisagentsAntipsychoticsBarbituratesCentral nervoussystem stimulantsNonsteroidal antiinflammatorydrugs(NSAIDs)Provide prescriber verified documentation <strong>of</strong> thenecessity <strong>of</strong> the duplication in the treatment plan.After 12 weeks (84 days) <strong>of</strong> concomitant oral andinjectable antipsychotic medication use, provideprescriber verified documentation <strong>of</strong> the necessity inthe treatment plan.Provide prescriber verified documentation <strong>of</strong> thenecessity <strong>of</strong> the duplication in the treatment plan.Provide prescriber verified documentation <strong>of</strong> thenecessity <strong>of</strong> the duplication in the treatment plan.After 60 days <strong>of</strong> concomitant use, provide prescriberverified documentation <strong>of</strong> the necessity <strong>of</strong> theduplication in the treatment plan.9. Status Change for Preferred Brand Name <strong>Drugs</strong>When the status <strong>of</strong> a previously preferred brand-name drug changes tononpreferred, pharmacies are given a transition period <strong>of</strong> up to 30 days toallow utilization <strong>of</strong> existing stock <strong>of</strong> the brand-name product.If additional stock remains beyond this period, pharmacies may call the Point<strong>of</strong> Sale (POS) Helpdesk at 877-463-7671 or 515-256-4608 (local) to requestan override for the nonpreferred brand-name drug with a recent statuschange.10. Travel or Vacation Supplies <strong>of</strong> MedicationRequests <strong>of</strong> medications for travel or vacation should be planned well inadvance <strong>of</strong> the departure date.The pharmacy can process the first month’s prescriptions as usual, and thenmay call the Point <strong>of</strong> Sale (POS) Helpdesk at 877-463-7671 or 515-256-4608(local) to obtain up to a one-month supply <strong>of</strong> medications to total up to a60-day supply <strong>of</strong> medication.