Brazilian Journal of Analytical Chemistry - BRJAC - Brazilian Journal ...
Brazilian Journal of Analytical Chemistry - BRJAC - Brazilian Journal ...
Brazilian Journal of Analytical Chemistry - BRJAC - Brazilian Journal ...
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
a m u l t iC o m m u t a t e d flow system f o r peraCetiC a C i d determination<br />
The sample consumption was 600 times lower for<br />
the flow system than for the reference method. The<br />
sampling throughput was 80 injections per hour, for<br />
the proposed system, which indicates that laboratory<br />
productivity would be enhanced by 3.6 or 18 times,<br />
compared to the Vacu-vials ® Kit and to the reference<br />
method, respectively.<br />
The performance <strong>of</strong> the multicommuted flow injection<br />
analysis process (MCFIA) in real sample analysis<br />
was evaluated using three disinfectant samples that<br />
were assayed using both the proposed procedure and<br />
the reference permanganate titration [12] method. The<br />
results are presented in Table III.<br />
taBlE iii. PEraCEtiC aCid C o n C E n t ra t i o n s (g l -1 ) in s a m P l E<br />
d i s i n fE C t a n t s o B t a i nE d By thE P ro P o s E d ProCEdurE (mCfia) a n d<br />
thE rEfErEnCE m E t h o d [12].<br />
sa m P l E s<br />
Pro P o s E d m E t h o d<br />
(g l -1 )<br />
rEfErEnCE m E t h o d<br />
(g l -1 )<br />
rE l a t i v E Er r o r<br />
(%)<br />
1 166.9 ± 16.4 163.4 ± 13.2 - 2.14<br />
2 169.8 ± 18.2 162.1 ± 4.8 - 4.75<br />
3 77.4 ± 6.4 101.3 ± 21.9 23.6<br />
The data reported indicate the presence <strong>of</strong> peracetic<br />
acid concentrations between 77 and 170 g L -1 with a<br />
RSD between 6.4 and 18.2 % using the method under<br />
study, compared with 4.8 to 21.9% for the reference<br />
method. The values <strong>of</strong> relative errors were both positive<br />
and negative, indicating that both procedures provide<br />
comparable results without systematic errors.<br />
In the reference method, the hydrogen peroxide<br />
present is first titrated under cold conditions with a<br />
standard solution <strong>of</strong> potassium permanganate. This<br />
titration eliminates the peroxide. Potassium iodide is<br />
then rapidly added and the iodine liberated by the<br />
peracid is titrated with standard thiosulphate solution<br />
[12]. In the proposed method there is no step to eliminate<br />
peroxide. As mentioned, the proposed method<br />
is based on the reaction <strong>of</strong> PAA with I - solution. The<br />
concentration <strong>of</strong> I - in the sample solution should be<br />
high enough to dissolve the I 2 resulting from the reaction<br />
<strong>of</strong> PAA (and H 2 O 2 ) with I - (reference 3). Probably,<br />
in sample 3, there was a high concentration H 2 O 2 and<br />
this reacted with I - generating a larger amount <strong>of</strong> I 2 .<br />
Thus, the excess <strong>of</strong> I - was not enough to solubilize I 2 ,<br />
so that the analytical signal was smaller, resulting in a<br />
lower PAA concentration.<br />
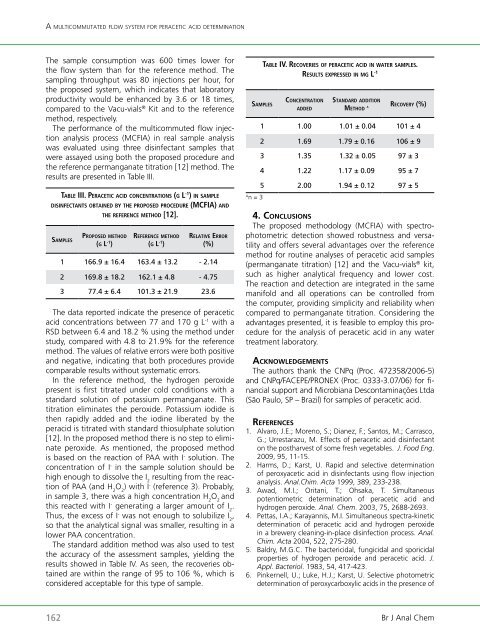
The standard addition method was also used to test<br />
the accuracy <strong>of</strong> the assessment samples, yielding the<br />
results showed in Table IV. As seen, the recoveries obtained<br />
are within the range <strong>of</strong> 95 to 106 %, which is<br />
considered acceptable for this type <strong>of</strong> sample.<br />
sa m P l E s<br />
taBlE iv. rECovEriEs o f PEraCEtiC aCid in w a t E r samPlEs.<br />
rEsults ExPrEssEd in m g l -1<br />
Co n C E n t r a t i o n<br />
162 Br J Anal Chem<br />
A n = 3<br />
a d d E d<br />
st a n d a rd a d d i t i o n<br />
mE t h o d a<br />
rECovEry (%)<br />
1 1.00 1.01 ± 0.04 101 ± 4<br />
2 1.69 1.79 ± 0.16 106 ± 9<br />
3 1.35 1.32 ± 0.05 97 ± 3<br />
4 1.22 1.17 ± 0.09 95 ± 7<br />
5 2.00 1.94 ± 0.12 97 ± 5<br />
4. co n c l u s Io n s<br />
The proposed methodology (MCFIA) with spectrophotometric<br />
detection showed robustness and versatility<br />
and <strong>of</strong>fers several advantages over the reference<br />
method for routine analyses <strong>of</strong> peracetic acid samples<br />
(permanganate titration) [12] and the Vacu-vials ® kit,<br />
such as higher analytical frequency and lower cost.<br />
The reaction and detection are integrated in the same<br />
manifold and all operations can be controlled from<br />
the computer, providing simplicity and reliability when<br />
compared to permanganate titration. Considering the<br />
advantages presented, it is feasible to employ this procedure<br />
for the analysis <strong>of</strong> peracetic acid in any water<br />
treatment laboratory.<br />
ac K n o w l e d g e m e n t s<br />
The authors thank the CNPq (Proc. 472358/2006-5)<br />
and CNPq/FACEPE/PRONEX (Proc. 0333-3.07/06) for financial<br />
support and Microbiana Descontaminações Ltda<br />
(São Paulo, SP – Brazil) for samples <strong>of</strong> peracetic acid.<br />
references<br />
1. Alvaro, J.E.; Moreno, S.; Dianez, F.; Santos, M.; Carrasco,<br />
G.; Urrestarazu, M. Effects <strong>of</strong> peracetic acid disinfectant<br />
on the postharvest <strong>of</strong> some fresh vegetables. J. Food Eng.<br />
2009, 95, 11-15.<br />
2. Harms, D.; Karst, U. Rapid and selective determination<br />
<strong>of</strong> peroxyacetic acid in disinfectants using flow injection<br />
analysis. Anal.Chim. Acta 1999, 389, 233-238.<br />
3. Awad, M.I.; Oritani, T.; Ohsaka, T. Simultaneous<br />
potentiometric determination <strong>of</strong> peracetic acid and<br />
hydrogen peroxide. Anal. Chem. 2003, 75, 2688-2693.<br />
4. Pettas, I.A.; Karayannis, M.I. Simultaneous spectra-kinetic<br />
determination <strong>of</strong> peracetic acid and hydrogen peroxide<br />
in a brewery cleaning-in-place disinfection process. Anal.<br />
Chim. Acta 2004, 522, 275-280.<br />
5. Baldry, M.G.C. The bactericidal, fungicidal and sporicidal<br />
properties <strong>of</strong> hydrogen peroxide and peracetic acid. J.<br />
Appl. Bacteriol. 1983, 54, 417-423.<br />
6. Pinkernell, U.; Luke, H.J.; Karst, U. Selective photometric<br />
determination <strong>of</strong> peroxycarboxylic acids in the presence <strong>of</strong>