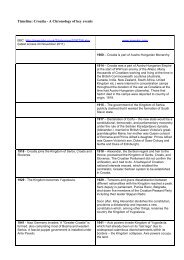
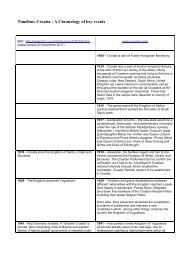
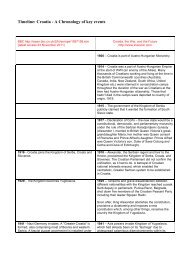
PUBLIC - Croatia, the War, and the Future
PUBLIC - Croatia, the War, and the Future
PUBLIC - Croatia, the War, and the Future
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
Glucose 5 %, Viaflo solution for infusion<br />
Glucose 5 % Viaflo solution for infusion<br />
Glucose 5% solution for infusion<br />
Glucose 5% solution for infusion<br />
Glucose 5% solution for infusion<br />
Glucose 5% Braun solution for infusion<br />
Glucose 5% Braun solution for infusion<br />
Glucose 5% Braun solution for infusion<br />
Glucose 5% Braun solution for infusion<br />
Glucose 5% Pliva solution for infusion<br />
Glucose 5% Pliva solution for infusion<br />
Glurenorm 30 mg tablets tablets<br />
GONAL-f 1050 IU/1.75 mL<br />
GONAL-f 300 IU/0.5 mL<br />
powder <strong>and</strong> diluent (in a<br />
syringe) for solution for<br />
injection<br />
solution for injection in<br />
pen syringe<br />
Republic of Irel<strong>and</strong><br />
Baxter S.A., Belgium;<br />
Baxter Healthcare Ltd,<br />
Great Britain; Bieffe<br />
Medital Sabinanigo,<br />
Spain; Baxter<br />
Healthcare S.A.,<br />
Republic of Irel<strong>and</strong><br />
Baxter S.A., Belgium;<br />
Baxter Healthcare Ltd,<br />
Great Britain; Bieffe<br />
Medital Sabinanigo,<br />
Spain; Baxter<br />
Healthcare S.A.,<br />
Republic of Irel<strong>and</strong><br />
Hrvatski zavod za<br />
transfuzijsku medicinu,<br />
Petrova 3, Zagreb,<br />
Republic of <strong>Croatia</strong><br />
Hrvatski zavod za<br />
transfuzijsku medicinu,<br />
Petrova 3, Zagreb,<br />
Republic of <strong>Croatia</strong><br />
Hrvatski zavod za<br />
transfuzijsku medicinu,<br />
Petrova 3, Zagreb,<br />
Republic of <strong>Croatia</strong><br />
B. Braun Melsungen<br />
AG, Carl-Braun-Straße<br />
1, Melsungen,<br />
Germany<br />
B. Braun Melsungen<br />
AG, Carl-Braun-Straße<br />
1, Melsungen,<br />
Germany<br />
B. Braun Melsungen<br />
AG, Carl-Braun-Straße<br />
1, Melsungen,<br />
Germany<br />
B. Braun Melsungen<br />
AG, Carl-Braun-Straße<br />
1, Melsungen,<br />
Germany<br />
Pliva <strong>Croatia</strong> Ltd., Ulica<br />
grada Vukovara 49,<br />
Zagreb, Republic of<br />
<strong>Croatia</strong><br />
Pliva <strong>Croatia</strong> Ltd., Ulica<br />
grada Vukovara 49,<br />
Zagreb, Republic of<br />
<strong>Croatia</strong><br />
Boehringer Ingelheim<br />
Ellas A.E., Atena,<br />
Greece<br />
Industria Farmaceutica<br />
Serono S.p.A., Bari,<br />
Italy<br />
Industria Farmaceutica<br />
Serono S.p.A., Bari,<br />
Italy<br />
50 plastic bags Viaflo with<br />
100 mL of solution for<br />
infusion, in a protective bag,<br />
supplied in a box<br />
20 plastic Viaflo bags with<br />
500 mL of infusion solution,<br />
in a protective bag, supplied<br />
in a box<br />
500 mL of solution in a glass<br />
bottle<br />
100 mL of solution in a glass<br />
bottle<br />
250-mL of solution in a glass<br />
bottle<br />
1000 mL of infusion solution<br />
in a plastic bottle (10 bottles<br />
in a box)<br />
250-mL of solution for<br />
infusion in a plastic bottle (10<br />
bottles in a box)<br />
500 mL of solution for<br />
infusion in a plastic bottle (10<br />
bottles in a box)<br />
100 mL of infusion solution<br />
in a plastic bottle (20 bottles<br />
in a box)<br />
500 mL of solution in a PVC<br />
bag, with a draining<br />
attachment (closed with<br />
combiflack stopper) <strong>and</strong> an<br />
application attachment, 12<br />
bags in a box<br />
500 mL of solution in a glass<br />
infusion bottle with a rubber<br />
stopper <strong>and</strong> a protective cap,<br />
10 bottles in a box<br />
60 (6x10) tablets in a white<br />
blister (PVC/PVDC//Al),<br />
supplied in a box<br />
Vial with powder <strong>and</strong> syringe<br />
with 2 mL of diluent, supplied<br />
in a box<br />
Pen-syringe with 0.5 mL of<br />
solution (in a 3-mL glass<br />
cartridge) <strong>and</strong> 5 needles in a<br />
plastic container, supplied in<br />
a box<br />
YES YES<br />
YES YES<br />
YES YES<br />
YES YES<br />
YES YES<br />
YES YES<br />
YES YES<br />
YES YES<br />
YES YES<br />
YES YES<br />
YES YES<br />
YES YES<br />
YES YES YES<br />
YES YES YES<br />
14509/11 WS/jj 292<br />
ANNEX II DGF 2A / DG K 2 EN