Report in English with a Dutch summary (KCE reports 63A)
Report in English with a Dutch summary (KCE reports 63A)
Report in English with a Dutch summary (KCE reports 63A)
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
108 Breast Cancer <strong>KCE</strong> <strong>reports</strong> 63<br />
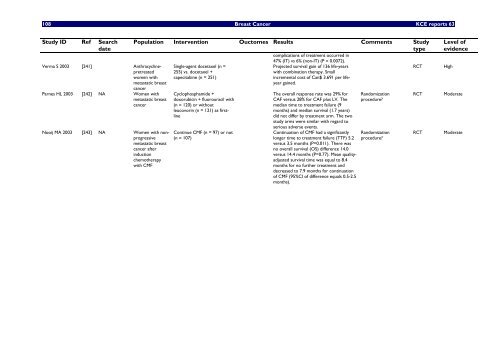
Study ID Ref Search<br />
date<br />
Verma S 2003 [241] Anthracycl<strong>in</strong>epretreated<br />
women <strong>with</strong><br />
metastatic breast<br />
Population Intervention Ouctomes Results Comments Study<br />
type<br />
cancer<br />
Parnes HL 2003 [242] NA Women <strong>with</strong><br />
metastatic breast<br />
cancer<br />
Nooij MA 2003 [243] NA Women <strong>with</strong> nonprogressive<br />
metastatic breast<br />
cancer after<br />
<strong>in</strong>duction<br />
chemotherapy<br />
<strong>with</strong> CMF<br />
S<strong>in</strong>gle-agent docetaxel (n =<br />
255) vs. docetaxel +<br />
capecitab<strong>in</strong>e (n = 251)<br />
Cyclophosphamide +<br />
doxorubic<strong>in</strong> + fluorouracil <strong>with</strong><br />
(n = 120) or <strong>with</strong>out<br />
leucovor<strong>in</strong> (n = 121) as firstl<strong>in</strong>e<br />
Cont<strong>in</strong>ue CMF (n = 97) or not<br />
(n = 107)<br />
complications of treatment occurred <strong>in</strong><br />
47% (IT) vs 6% (non-IT) (P = 0.0072).<br />
Projected survival ga<strong>in</strong> of 136 life-years<br />
<strong>with</strong> comb<strong>in</strong>ation therapy. Small<br />
<strong>in</strong>cremental cost of Can$ 3.691 per lifeyear<br />
ga<strong>in</strong>ed.<br />
The overall response rate was 29% for<br />
CAF versus 28% for CAF plus LV. The<br />
median time to treatment failure (9<br />
months) and median survival (1.7 years)<br />
did not differ by treatment arm. The two<br />
study arms were similar <strong>with</strong> regard to<br />
serious adverse events.<br />
Cont<strong>in</strong>uation of CMF had a significantly<br />
longer time to treatment failure (TTF) 5.2<br />
versus 3.5 months (P=0.011). There was<br />
no overall survival (OS) difference 14.0<br />
versus 14.4 months (P=0.77). Mean qualityadjusted<br />
survival time was equal to 8.4<br />
months for no further treatment and<br />
decreased to 7.9 months for cont<strong>in</strong>uation<br />
of CMF (95%CI of difference equals 0.5-2.5<br />
months).<br />
Randomization<br />
procedure?<br />
Randomization<br />
procedure?<br />
RCT High<br />
Level of<br />
evidence<br />
RCT Moderate<br />
RCT Moderate