Thesis for the Degree of Doctor of Philosophy - DTU Orbit
Thesis for the Degree of Doctor of Philosophy - DTU Orbit
Thesis for the Degree of Doctor of Philosophy - DTU Orbit
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
1. Introduction<br />
1.1 Iodine in nature<br />
1.1.1 Stable iodine ( 127 I)<br />
In 1811 <strong>the</strong> French chemist Bernard Courtois discovered iodine by a <strong>for</strong>tuitous accident when he sublimed<br />
<strong>the</strong> element from seaweed ash with sulphuric acid. The name comes from a Greek word <strong>for</strong> "violet colored".<br />
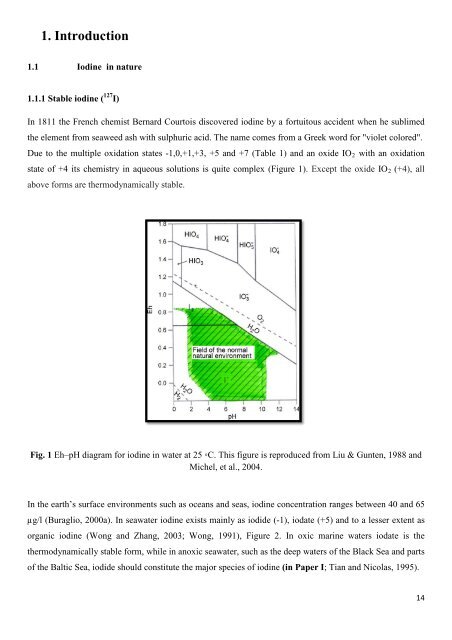
Due to <strong>the</strong> multiple oxidation states -1,0,+1,+3, +5 and +7 (Table 1) and an oxide IO2 with an oxidation<br />
state <strong>of</strong> +4 its chemistry in aqueous solutions is quite complex (Figure 1). Except <strong>the</strong> oxide IO2 (+4), all<br />
above <strong>for</strong>ms are <strong>the</strong>rmodynamically stable.<br />
Fig. 1 Eh–pH diagram <strong>for</strong> iodine in water at 25 ◦C. This figure is reproduced from Liu & Gunten, 1988 and<br />
Michel, et al., 2004.<br />
In <strong>the</strong> earth’s surface environments such as oceans and seas, iodine concentration ranges between 40 and 65<br />
µg/l (Buraglio, 2000a). In seawater iodine exists mainly as iodide (-1), iodate (+5) and to a lesser extent as<br />
organic iodine (Wong and Zhang, 2003; Wong, 1991), Figure 2. In oxic marine waters iodate is <strong>the</strong><br />
<strong>the</strong>rmodynamically stable <strong>for</strong>m, while in anoxic seawater, such as <strong>the</strong> deep waters <strong>of</strong> <strong>the</strong> Black Sea and parts<br />
<strong>of</strong> <strong>the</strong> Baltic Sea, iodide should constitute <strong>the</strong> major species <strong>of</strong> iodine (in Paper I; Tian and Nicolas, 1995).<br />
14