Thesis for the Degree of Doctor of Philosophy - DTU Orbit
Thesis for the Degree of Doctor of Philosophy - DTU Orbit
Thesis for the Degree of Doctor of Philosophy - DTU Orbit
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
numbers 11-14), which is about <strong>the</strong> same as <strong>the</strong> experiments above using 0.01M K2S2O8 (Table 9). These<br />
findings might be attributed to <strong>the</strong> sample matrix (organic matter), which may partially precipitate when<br />
adding silver nitrate, and thus <strong>the</strong> iodide, iodate and organic iodine were precipitated toge<strong>the</strong>r. As long as<br />
large molecular weight humic molecules are <strong>the</strong> main carrier <strong>of</strong> organic iodine species direct co-<br />
precipitation without previous oxidation <strong>of</strong> <strong>the</strong> sample may thus be an alternative method. In fresh water<br />
highly enriched in organic matter (e.g. Humic substances) <strong>the</strong> increased ion strength due to <strong>the</strong> addition <strong>of</strong><br />
<strong>the</strong> chemicals used induce a visible coagulation and co-precipitation <strong>of</strong> organic matter.<br />
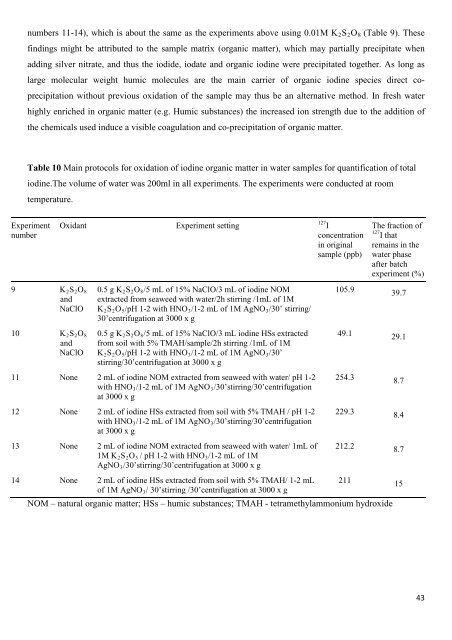
Table 10 Main protocols <strong>for</strong> oxidation <strong>of</strong> iodine organic matter in water samples <strong>for</strong> quantification <strong>of</strong> total<br />
iodine.The volume <strong>of</strong> water was 200ml in all experiments. The experiments were conducted at room<br />
temperature.<br />
Experiment<br />
number<br />
9 K2S2O8<br />
and<br />
NaClO<br />
10 K2S2O8<br />
and<br />
NaClO<br />
Oxidant Experiment setting<br />
0.5 g K2S2O8/5 mL <strong>of</strong> 15% NaClO/3 mL <strong>of</strong> iodine NOM<br />
extracted from seaweed with water/2h stirring /1mL <strong>of</strong> 1M<br />
K2S2O5/pH 1-2 with HNO3/1-2 mL <strong>of</strong> 1M AgNO3/30’ stirring/<br />
30’centrifugation at 3000 x g<br />
0.5 g K2S2O8/5 mL <strong>of</strong> 15% NaClO/3 mL iodine HSs extracted<br />
from soil with 5% TMAH/sample/2h stirring /1mL <strong>of</strong> 1M<br />
K2S2O5/pH 1-2 with HNO3/1-2 mL <strong>of</strong> 1M AgNO3/30’<br />
stirring/30’centrifugation at 3000 x g<br />
11 None 2 mL <strong>of</strong> iodine NOM extracted from seaweed with water/ pH 1-2<br />
with HNO3/1-2 mL <strong>of</strong> 1M AgNO3/30’stirring/30’centrifugation<br />
at 3000 x g<br />
12 None 2 mL <strong>of</strong> iodine HSs extracted from soil with 5% TMAH / pH 1-2<br />
with HNO3/1-2 mL <strong>of</strong> 1M AgNO3/30’stirring/30’centrifugation<br />
at 3000 x g<br />
13 None 2 mL <strong>of</strong> iodine NOM extracted from seaweed with water/ 1mL <strong>of</strong><br />
1M K2S2O5 / pH 1-2 with HNO3/1-2 mL <strong>of</strong> 1M<br />
AgNO3/30’stirring/30’centrifugation at 3000 x g<br />
127 I<br />
concentration<br />
in original<br />
sample (ppb)<br />
The fraction <strong>of</strong><br />
127 I that<br />
remains in <strong>the</strong><br />
water phase<br />
after batch<br />
experiment (%)<br />
105.9 39.7<br />
49.1 29.1<br />
254.3 8.7<br />
229.3 8.4<br />
212.2 8.7<br />
14 None 2 mL <strong>of</strong> iodine HSs extracted from soil with 5% TMAH/ 1-2 mL<br />
<strong>of</strong> 1M AgNO3/ 30’stirring /30’centrifugation at 3000 x g<br />
211 15<br />
NOM – natural organic matter; HSs – humic substances; TMAH - tetramethylammonium hydroxide<br />
43