CHAPTER 3 Tumours of the Stomach - Pathology Outlines
CHAPTER 3 Tumours of the Stomach - Pathology Outlines
CHAPTER 3 Tumours of the Stomach - Pathology Outlines
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
A<br />
B<br />
C<br />
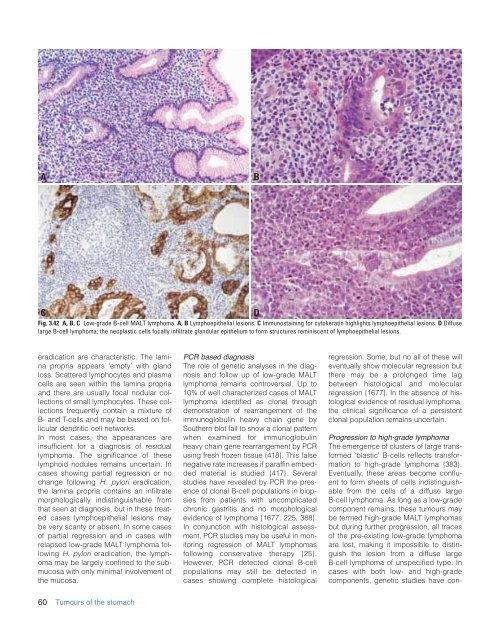
Fig. 3.42 A, B, C Low-grade B-cell MALT lymphoma. A, B Lymphoepi<strong>the</strong>lial lesions. C Immunostaining for cytokeratin highlights lymphoepi<strong>the</strong>lial lesions. D Diffuse<br />
large B-cell lymphoma; <strong>the</strong> neoplastic cells focally infiltrate glandular epi<strong>the</strong>lium to form structures reminiscent <strong>of</strong> lymphoepi<strong>the</strong>lial lesions.<br />
D<br />
eradication are characteristic. The lamina<br />
propria appears ‘empty’ with gland<br />
loss. Scattered lymphocytes and plasma<br />
cells are seen within <strong>the</strong> lamina propria<br />
and <strong>the</strong>re are usually focal nodular collections<br />
<strong>of</strong> small lymphocytes. These collections<br />
frequently contain a mixture <strong>of</strong><br />
B- and T-cells and may be based on follicular<br />
dendritic cell networks.<br />
In most cases, <strong>the</strong> appearances are<br />
insufficient for a diagnosis <strong>of</strong> residual<br />
lymphoma. The significance <strong>of</strong> <strong>the</strong>se<br />
lymphoid nodules remains uncertain. In<br />
cases showing partial regression or no<br />
change following H. pylori eradication,<br />
<strong>the</strong> lamina propria contains an infiltrate<br />
morphologically indistinguishable from<br />
that seen at diagnosis, but in <strong>the</strong>se treated<br />
cases lymphoepi<strong>the</strong>lial lesions may<br />
be very scanty or absent. In some cases<br />
<strong>of</strong> partial regression and in cases with<br />
relapsed low-grade MALT lymphoma following<br />
H. pylori eradication, <strong>the</strong> lymphoma<br />
may be largely confined to <strong>the</strong> submucosa<br />
with only minimal involvement <strong>of</strong><br />
<strong>the</strong> mucosa.<br />
PCR based diagnosis<br />
The role <strong>of</strong> genetic analyses in <strong>the</strong> diagnosis<br />
and follow up <strong>of</strong> low-grade MALT<br />
lymphoma remains controversial. Up to<br />
10% <strong>of</strong> well characterized cases <strong>of</strong> MALT<br />
lymphoma identified as clonal through<br />
demonstration <strong>of</strong> rearrangement <strong>of</strong> <strong>the</strong><br />
immunoglobulin heavy chain gene by<br />
Sou<strong>the</strong>rn blot fail to show a clonal pattern<br />
when examined for immunoglobulin<br />
heavy chain gene rearrangement by PCR<br />
using fresh frozen tissue {418}. This false<br />
negative rate increases if paraffin embedded<br />
material is studied {417}. Several<br />
studies have revealed by PCR <strong>the</strong> presence<br />
<strong>of</strong> clonal B-cell populations in biopsies<br />
from patients with uncomplicated<br />
chronic gastritis and no morphological<br />
evidence <strong>of</strong> lymphoma {1677, 225, 388}.<br />
In conjunction with histological assessment,<br />
PCR studies may be useful in monitoring<br />
regression <strong>of</strong> MALT lymphomas<br />
following conservative <strong>the</strong>rapy {25}.<br />
However, PCR detected clonal B-cell<br />
populations may still be detected in<br />
cases showing complete histological<br />
regression. Some, but no all <strong>of</strong> <strong>the</strong>se will<br />
eventually show molecular regression but<br />
<strong>the</strong>re may be a prolonged time lag<br />
between histological and molecular<br />
regression {1677}. In <strong>the</strong> absence <strong>of</strong> histological<br />
evidence <strong>of</strong> residual lymphoma,<br />
<strong>the</strong> clinical significance <strong>of</strong> a persistent<br />
clonal population remains uncertain.<br />
Progression to high-grade lymphoma<br />
The emergence <strong>of</strong> clusters <strong>of</strong> large transformed<br />
‘blastic’ B-cells reflects transformation<br />
to high-grade lymphoma {383}.<br />
Eventually, <strong>the</strong>se areas become confluent<br />
to form sheets <strong>of</strong> cells indistinguishable<br />
from <strong>the</strong> cells <strong>of</strong> a diffuse large<br />
B-cell lymphoma. As long as a low-grade<br />
component remains, <strong>the</strong>se tumours may<br />
be termed high-grade MALT lymphomas<br />
but during fur<strong>the</strong>r progression, all traces<br />
<strong>of</strong> <strong>the</strong> pre-existing low-grade lymphoma<br />
are lost, making it impossible to distinguish<br />
<strong>the</strong> lesion from a diffuse large<br />
B-cell lymphoma <strong>of</strong> unspecified type. In<br />
cases with both low- and high-grade<br />
components, genetic studies have con-<br />
60 <strong>Tumours</strong> <strong>of</strong> <strong>the</strong> stomach