Topic 1 - Matter and Energy - Revsworld
Topic 1 - Matter and Energy - Revsworld
Topic 1 - Matter and Energy - Revsworld
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
.<br />
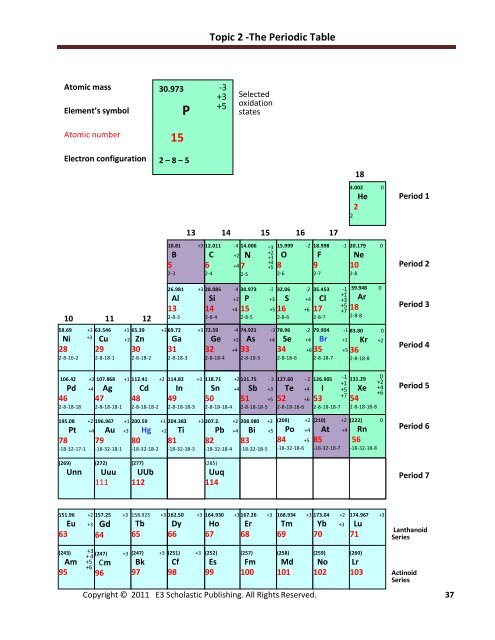
<strong>Topic</strong> 2 -The Periodic Table<br />
Atomic mass<br />
Element’s symbol<br />
30.973<br />
P<br />
-3<br />
+3<br />
+5<br />
Selected<br />
oxidation<br />
states<br />
Atomic number<br />
15<br />
Electron configuration<br />
2 – 8 – 5<br />
18<br />
4.002 0<br />
He<br />
2<br />
2<br />
Period 1<br />
10 11 12<br />
58.69 +2<br />
Ni<br />
+3<br />
28<br />
2-8-16-2<br />
63.546 +1<br />
Cu +2<br />
29<br />
2-8-18-1<br />
65.39 +2<br />
Zn<br />
30<br />
2-8-18-2<br />
10.81 +3<br />
B<br />
5<br />
2-3<br />
26.981 +3<br />
Al<br />
13<br />
2-8-3<br />
69.72 +3<br />
Ga<br />
31<br />
2-8-18-3<br />
13 14 15 16 17<br />
12.011 -4<br />
C +2<br />
6 +4<br />
2-4<br />
28.085 -4<br />
Si +2<br />
14 +4<br />
2-8-4<br />
14.006<br />
N<br />
7<br />
2-5<br />
-3<br />
+2<br />
+3<br />
+4<br />
+5<br />
30.973 -3<br />
P +3<br />
15 +5<br />
2-8-5<br />
15.999 -2<br />
O<br />
8<br />
2-6<br />
32.06 -2<br />
S +4<br />
16 +6<br />
2-8-6<br />
18.998 -1<br />
F<br />
9<br />
2-7<br />
35.453<br />
Cl<br />
17<br />
2-8-7<br />
-1<br />
+1<br />
+3<br />
+5<br />
+7<br />
20.179 0<br />
Ne<br />
10<br />
2-8<br />
39.948 0<br />
Ar<br />
18<br />
2-8-8<br />
72.59 -4 69.72 74.921 +3 -3<br />
78.96 -2 79.904 -1<br />
Ge +2 Ga As +4<br />
Se +4 Br +1<br />
32 +4<br />
31 33<br />
34 +6 35 +5 36<br />
2-8-18-4 2-8-18-3 2-8-18-5<br />
2-8-18-6 2-8-18-7 2-8-18-8<br />
83.80 0<br />
Kr +2<br />
Period 2<br />
Period 3<br />
Period 4<br />
106.42 +2<br />
Pd +4<br />
46<br />
2-8-18-18<br />
195.08 +2<br />
Pt +4<br />
78<br />
-18-32-17-1<br />
107.868 +1<br />
Ag<br />
47<br />
2-8-18-18-1<br />
196.967 +1<br />
Au +3<br />
79<br />
-18-32-18-1<br />
112.41 +2<br />
Cd<br />
48<br />
2-8-18-18-2<br />
200.59 +1<br />
Hg +2<br />
80<br />
-18-32-18-2<br />
114.82 +3<br />
In<br />
49<br />
2-8-18-18-3<br />
204.383 +3<br />
Ti<br />
81<br />
-18-32-18-3<br />
118.71 +2 114.82 121.75 - +3 3<br />
Sn +4 In Sb +3<br />
50 49 51 +5<br />
2-8-18-18-4 2-8-18-18-3<br />
2-8-18-18-5<br />
207.2. +2<br />
Pb +4<br />
82<br />
-18-32-18-4<br />
208.980 +3<br />
Bi +5<br />
83<br />
-18-32-18-5<br />
127.60 - 2<br />
Te +4<br />
52 +6<br />
2-8-18-18-6<br />
126.905<br />
I<br />
53<br />
2-8-18-18-7<br />
-1<br />
+1<br />
+5<br />
+7<br />
(209) +2<br />
(210) +2<br />
Po +4<br />
At +4 +4<br />
84 +5<br />
85<br />
-18-32-18-6<br />
-18-32-18-7<br />
131.29<br />
Xe<br />
54<br />
2-8-18-18-8<br />
0<br />
+2<br />
+4<br />
+6<br />
(222) 0<br />
Rn<br />
56<br />
-18-32-18-8<br />
Period 5<br />
Period 6<br />
(269)<br />
Unn<br />
(272)<br />
Uuu<br />
111<br />
(277)<br />
UUb<br />
112<br />
(285)<br />
Uuq<br />
114<br />
Period 7<br />
151.96 +2<br />
Eu +3<br />
63<br />
157.25 +3<br />
Gd<br />
64<br />
158.925 +3<br />
Tb<br />
65<br />
162.50 +3<br />
Dy<br />
66<br />
164.930 +3<br />
Ho<br />
67<br />
167.26 +3<br />
Er<br />
68<br />
168.934 +3 173.04 +2<br />
Tm Yb +3<br />
69 70<br />
174.967 +3<br />
Lu<br />
71<br />
Lanthanoid<br />
Series<br />
(243)<br />
Am<br />
95<br />
+3<br />
+ 4<br />
+5<br />
+6<br />
(247) +3<br />
Cm<br />
96<br />
(247) +3<br />
Bk<br />
97<br />
(251) +3<br />
Cf<br />
98<br />
(252)<br />
Es<br />
99<br />
(257)<br />
Fm<br />
100<br />
(258)<br />
Md<br />
101<br />
Copyright © 2011 E3 Scholastic Publishing. All Rights Reserved. 37<br />
(259)<br />
No<br />
102<br />
(260)<br />
Lr<br />
103<br />
Actinoid<br />
Series