teaching - Earth Science Teachers' Association
teaching - Earth Science Teachers' Association
teaching - Earth Science Teachers' Association
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
TEACHING EARTH SCIENCES ● Volume 26 ● Number 3, 2001<br />
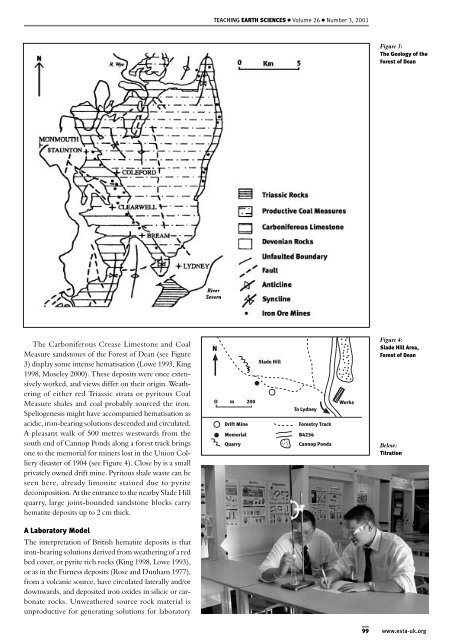
Figure 3:<br />
The Geology of the<br />
Forest of Dean<br />
The Carboniferous Crease Limestone and Coal<br />
Measure sandstones of the Forest of Dean (see Figure<br />
3) display some intense hematisation (Lowe 1993, King<br />
1998, Moseley 2000). These deposits were once extensively<br />
worked, and views differ on their origin. Weathering<br />
of either red Triassic strata or pyritous Coal<br />
Measure shales and coal probably sourced the iron.<br />
Speliogenesis might have accompanied hematisation as<br />
acidic, iron-bearing solutions descended and circulated.<br />
A pleasant walk of 500 metres westwards from the<br />
south end of Cannop Ponds along a forest track brings<br />
one to the memorial for miners lost in the Union Colliery<br />
disaster of 1904 (see Figure 4). Close by is a small<br />
privately owned drift mine. Pyritous shale waste can be<br />
seen here, already limonite stained due to pyrite<br />
decomposition. At the entrance to the nearby Slade Hill<br />
quarry, large joint-bounded sandstone blocks carry<br />
hematite deposits up to 2 cm thick.<br />
N<br />
O m 200<br />
Drift Mine<br />
Memorial<br />
Quarry<br />
Slade Hill<br />
To Lydney<br />
Forestry Track<br />
B4234<br />
Cannop Ponds<br />
Works<br />
Figure 4:<br />
Slade Hill Area,<br />
Forest of Dean<br />
Below:<br />
Titration<br />
A Laboratory Model<br />
The interpretation of British hematite deposits is that<br />
iron-bearing solutions derived from weathering of a red<br />
bed cover, or pyrite rich rocks (King 1998, Lowe 1993),<br />
or as in the Furness deposits (Rose and Dunham 1977),<br />
from a volcanic source, have circulated laterally and/or<br />
downwards, and deposited iron oxides in silicic or carbonate<br />
rocks. Unweathered source rock material is<br />
unproductive for generating solutions for laboratory<br />
99 www.esta-uk.org