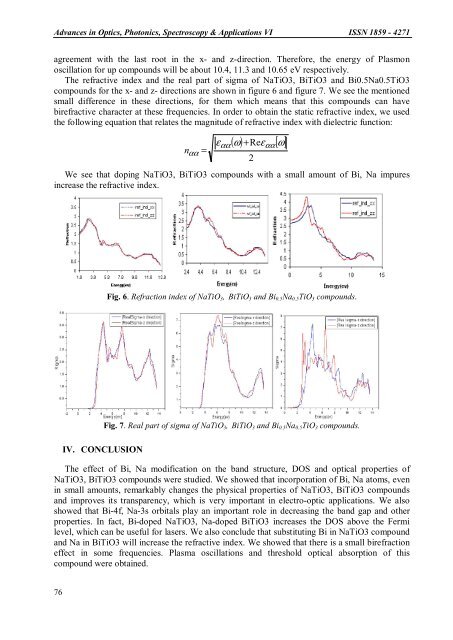
Advances in Optics, Photonics, Spectroscopy & Applications <strong>VI</strong> <strong>ISSN</strong> <strong>1859</strong> - <strong>4271</strong>agreement with the last root in the x- and z-direction. Therefore, the energy of Plasmonoscillation for up compounds will be about 10.4, 11.3 and 10.65 eV respectively.The refractive index and the real part of sigma of NaTiO3, BiTiO3 and Bi0.5Na0.5TiO3compounds for the x- and z- directions are shown in figure 6 and figure 7. We see the mentionedsmall difference in these directions, for them which means that this compounds can havebirefractive character at these frequencies. In order to obtain the static refractive index, we usedthe followi<strong>ng</strong> equation that relates the magnitude of refractive index with dielectric function:( ω) + Reε[ ω]εααααn αα =2We see that dopi<strong>ng</strong> NaTiO3, BiTiO3 compounds with a small amount of Bi, Na impuresincrease the refractive index.Fig. 6. Refraction index of NaTiO 3 , BiTiO 3 and Bi 0.5 Na 0.5 TiO 3 compounds.Fig. 7. Real part of sigma of NaTiO 3 , BiTiO 3 and Bi 0.5 Na 0.5 TiO 3 compounds.IV. CONCLUSIONThe effect of Bi, Na modification on the band structure, DOS and optical properties ofNaTiO3, BiTiO3 compounds were studied. We showed that incorporation of Bi, Na atoms, evenin small amounts, remarkably cha<strong>ng</strong>es the <stro<strong>ng</strong>>ph</stro<strong>ng</strong>>ysical properties of NaTiO3, BiTiO3 compoundsand improves its transparency, which is very important in electro-optic applications. We alsoshowed that Bi-4f, Na-3s orbitals play an important role in decreasi<strong>ng</strong> the band gap and otherproperties. In fact, Bi-doped NaTiO3, Na-doped BiTiO3 increases the DOS above the Fermilevel, which can be useful for lasers. We also conclude that substitu<s<stro<strong>ng</strong>>tro<strong>ng</strong></stro<strong>ng</strong>>>tin</s<stro<strong>ng</strong>>tro<strong>ng</strong></stro<strong>ng</strong>>>g Bi in NaTiO3 compoundand Na in BiTiO3 will increase the refractive index. We showed that there is a small birefractioneffect in some frequencies. Plasma oscillations and threshold optical absorption of thiscompound were obtained.76
Nhữ<strong>ng</strong> tiến bộ <stro<strong>ng</strong>>tro<strong>ng</strong></stro<strong>ng</strong>> <stro<strong>ng</strong>>Qua<strong>ng</strong></stro<strong>ng</strong>> học, <stro<strong>ng</strong>>Qua<strong>ng</strong></stro<strong>ng</strong>> <stro<strong>ng</strong>>ph</stro<strong>ng</strong>>ổ và Ứ<strong>ng</strong> dụ<strong>ng</strong> <strong>VI</strong> <strong>ISSN</strong> <strong>1859</strong> - <strong>4271</strong>Compound Method Energy gapNaTiO 3BiTiO 3BiTiO 3GGAGGAExp.2.52.62.8 (20)REFERENCES[1] G. A. Smolenskii, A. I. Agraovskaya, Dielectric polarization of a number of complex compounds,Sov. Phys. Solid State 1. pp. 1429-1437 1959.[2] G. A. Smolenskii, V. A. Isupv, A. I. Afranovskaya, N. N. Krainik, New ferroelectrics of complexcomposition. IV, Sov. Solid State (E<strong>ng</strong>l. Transl.) 2 . pp. 2651-2654 1961.[3] K. Sakata, Y. Masuda, Ferroelectric and antiferroelectric properties of (Na 0.5 Bi 0.5 )TiO 3 -SrTiO 3 solidsolution ceramics, Ferroelectrics 7. pp.347-349 1974.[4] V. A. Isupov, I. P. Pronin, T. V. Kruzin, Temperature dependence of birefri<strong>ng</strong>ence and opalescence ofthe sodium bismuth titanate crystals, Ferroelectr. Lett. 2. pp. 205-208 1984.[5] I. P. Pronin, P. P. Syrnikov, V. A. Isupov, V. M. Egorov, N. V. Zaitseva, Peculiarities of <stro<strong>ng</strong>>ph</stro<strong>ng</strong>>asetransitions in sodium-bismuth titanate, Ferroelectrics 25. pp. 395-397 1980.[6] S. Kuharua<strong>ng</strong>ro<strong>ng</strong>, W. A. Schulze Characterization of Bi 0.5 Na 0.5 TiO 3 -PbTiO 3 dielectric materials, J.Am. Ceram. Soc. 79. pp. 1273-1280 1996.[7] Y. Lin, S. Zhao, N. Cai, J. Wu, X. Zhou, C. W. Nan, Effects of dopi<strong>ng</strong> Eu 2 O 3 on the <stro<strong>ng</strong>>ph</stro<strong>ng</strong>>asetransformation and piezoelectric properties of Bi 0.5 Na 0.5 TiO 3 -based ceramics, Materials Sciense andE<strong>ng</strong>ineeri<strong>ng</strong> B 99. pp. 449-452 2003.[8] H. Nagata, T. Takenaka, Additive effects on electrical properties of ( Bi 0.5 Na 0.5 ) TiO 3 ferroelectricceramic, Journal of the European Ceramic Society 23. pp. 1299-1302 2001.[9] A. Ioachim, M. G. Banciu, L. Nedelcu, H. Alexandru, C. Berbecaru, D. Ghetu, G. Stoica, BNTceramics synthesis and characterization, Materials Science and E<strong>ng</strong>ineeri<strong>ng</strong> B 109. pp. 183-1872004.[10] A. Herabut, A. Safari, Effects of substitution in A-and B-site cations of Bi 0.5 Na 0.5 TiO 3 Proceedi<strong>ng</strong>sof the Tenth IEEE International Symposium, 1996, pp. 775-778.[11] Hardee KL, Bard AJ. Semiconductor electrode. J. Electrochem Soc 124-215 1977.[12] Thaninimulla CTK, Takata T, Hara M, Kondo J, Domen K, Effect of chromium addition for<stro<strong>ng</strong>>ph</stro<strong>ng</strong>>otocatalytic overall water split<s<stro<strong>ng</strong>>tro<strong>ng</strong></stro<strong>ng</strong>>>tin</s<stro<strong>ng</strong>>tro<strong>ng</strong></stro<strong>ng</strong>>>g on Ni-K 2 La 2 Ti 3 O 10 . J Catal pp. 196-362 2000.[13] C. G. Macarovici, G. Morar, Revue Roumiane de Chimie 17. pp. 1051-1059 1972.[14] J. T. Dawley, R. Radspinner, B. J. J. Zelinski, D. R. Uhlmann, Journal of Sol-Gel Science andTechnology 20. pp. 85-93 2001.[15] Kohn W and Sham L J 1965 Phys. Rev. 140A 1133.[16] BLAHA P, Schwarz K, Madsen G K H, Kvasnicka D and Luitz J WIEN2k, An A ugmented PlaneWave Plus Local Orbital Program for Calcula<s<stro<strong>ng</strong>>tro<strong>ng</strong></stro<strong>ng</strong>>>tin</s<stro<strong>ng</strong>>tro<strong>ng</strong></stro<strong>ng</strong>>>g Crystal Properties (Vienna, Austria: ViennaUniversity of Technology)[17] K. Schwarz, P. Blaha and G. K. H. Madsen Physics Communi Ca<s<stro<strong>ng</strong>>tro<strong>ng</strong></stro<strong>ng</strong>>>tin</s<stro<strong>ng</strong>>tro<strong>ng</strong></stro<strong>ng</strong>>>s. pp. 1-6 2002.[18] Perdew J. P, Burke S and Ernzerhof M 1996 Generalized gradient approximation made simple Phys.Rev. Lett. 7 3865.[19] de. L. Kronig R. 1926 On the theory of the dispersion of x-rays J. Opt. Soc. Am. 12. pp. 547-57.[20] C. Jovalekic, M. Zdujic, Lj. Atanasoska, " Surface analysis of bismuth titanate by Auger and X-ray<stro<strong>ng</strong>>ph</stro<strong>ng</strong>>otoelectron spectroscopy " ; Journal of Alloys and Compounds 469. pp. 441-444 2009.77