Table 2— AASM Levels of Re<strong>com</strong>mendationsStrength of Re<strong>com</strong>mendationStandardGuidelineOptionDefinitionThis is a generally accepted patient-care strategy that reflects a high degree of clinical certainty.The term standard generally implies the use of Level 1 Evidence, which directly addresses theclinical issue, or overwhelming Level 2 Evidence.This is a patient-care strategy that reflects a moderate degree of clinical certainty. The term guidelineimplies the use of Level 2 Evidence or a consensus of Level 3 Evidence.This is a patient-care strategy that which reflects uncertain clinical use. The term option implieseither inconclusive or conflicting evidence or conflicting expert opinion.as new information be<strong>com</strong>es available. Each article entered in theevidence tables of the <strong>com</strong>panion review paper was evaluated usingthe Standards of Practice Committee’s levels of evidence (Table1). This evidence is used to support the strength of the re<strong>com</strong>mendations(Table 2) in this paper. Square-bracketed numbers in thispaper refer to sections, tables, or references in the ac<strong>com</strong>panyingreview papers. Other citations, noted by superscripted numbers, referto the reference list at the end of this paper.3.0 RESULTS AND RECOMMENDATIONSThe following re<strong>com</strong>mendations reflect the evidence regardingthe diagnosis and treatment of CRSDs in clinical practice obtainedfrom the two ac<strong>com</strong>panying reviews. For brevity, the findings andre<strong>com</strong>mendations are summarized in Table 3. Specific details arereviewed in the practice parameters below.3.1 General Re<strong>com</strong>mendations for Evaluation of Circadian RhythmSleep DisordersThis re<strong>com</strong>mendation is unchanged from the recently updatedpractice parameter paper on the use of actigraphy. 6 This priorpractice parameter paper and ac<strong>com</strong>panying review provided evidencethat changes in actigraphy measures are in agreement withother out<strong>com</strong>e measures in the assessment of response to interventionin patients with CRSDs.3.1.4 There is insufficient evidence to re<strong>com</strong>mend the routine useof the Morningness-Eveningness Questionnaire (MEQ) for theclinical evaluation of CRSDs. (Option)Information regarding evidence for utility of MEQ in specificCRSDs is discussed under the disorder headings in the sectionbelow.3.1.5 Circadian phase markers are useful to determine circadianphase and confirm the diagnosis of FRD in sighted and unsightedpatients but there is insufficient evidence to re<strong>com</strong>mend their routineuse in the diagnosis of SWD, JLD, ASPD, DSPD, or ISWR. (Option)Information regarding evidence for specific CRSDs is discussedunder the disorder headings in the section below.3.1.1 Use of a sleep log or diary is indicated in the assessment ofpatients with a suspected CRSD. (Guideline)This re<strong>com</strong>mendation was determined by inclusion of the useof sleep logs in the International Classification of Sleep Disorders,2 nd Edition (ICSD-2) 5 diagnostic criteria for all CRSDs exceptjet lag. This re<strong>com</strong>mendation was additionally supported byconsensus opinion of the AASM SPC <strong>com</strong>mittee.3.1.2 Actigraphy is indicated to assist in evaluation of patientssuspected of CRSDs, including irregular sleep-wake disorder(ISWR), free-running disorder (FRD) (with or without blindness)(Option), and in advanced sleep phase disorder (ASPD), delayedsleep phase disorder (DSPD), and shift work disorder (SWD).(Guideline)This re<strong>com</strong>mendation reiterates the recently updated practice parameterpaper on the use of actigraphy. 6 Here, we indicate specificdisorders. There is generally good agreement among studies showingthat actigraphy data correlate with polysomnography (whenused), sleep logs, and markers of circadian phase in patients withcircadian rhythm sleep disorders, with the conditions indicated.3.1.3 Actigraphy is useful as an out<strong>com</strong>e measure in evaluatingthe response to treatment for CRSDs. (Guideline)SLEEP, Vol. 30, No. 11, 2007 14473.1.6 Polysomnography is indicated to rule out another primarysleep disorder in patients with symptoms suggestive of both aCRSD and another primary sleep disorder, but is not routinelyindicated for the diagnosis of CRSDs. (Standard)This re<strong>com</strong>mendation reiterates the recently updated practiceparameter paper on the indications for polysomnography andrelated procedures. 10 Polysomnography may be indicated whenconsidering a diagnosis of a CRSD to exclude other potentialcauses for sleep related <strong>com</strong>plaints. For example, shift workerswith hypersomnia may have both suspected obstructive sleepapnea and clinical characteristics consistent with shift work disorder.In this event, PSG is indicated to evaluate and establishappropriate therapy for OSA.3.2 Re<strong>com</strong>mendations for Evaluation and Treatments of CircadianRhythm Sleep Disorders3.2.1 Shift Work DisorderShift work refers to non-standard work schedules, includingpermanent or intermittent night work, early morning work, androtating schedules. An estimated 20% of U.S. workers are involvedin some form of shift work. The percentage of workerswho meet criteria for the diagnosis of shift work disorder (SWD)(i.e., development of sleep disturbances and impairment of wakingalertness and performance) is unclear, and there appear tobe individual differences in susceptibility to SWD (phase tolerance).Practice Parameters for the Clinical Evaluation of CRSD—Morgenthaler et al
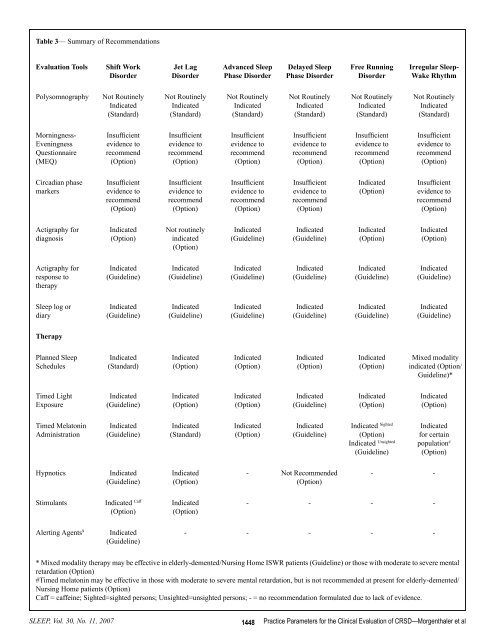
Table 3— Summary of Re<strong>com</strong>mendationsEvaluation ToolsShift WorkDisorderJet LagDisorderAdvanced SleepPhase DisorderDelayed SleepPhase DisorderFree RunningDisorderIrregular Sleep-Wake RhythmPolysomnographyNot RoutinelyIndicated(Standard)Not RoutinelyIndicated(Standard)Not RoutinelyIndicated(Standard)Not RoutinelyIndicated(Standard)Not RoutinelyIndicated(Standard)Not RoutinelyIndicated(Standard)Morningness-EveningnessQuestionnaire(MEQ)Insufficientevidence tore<strong>com</strong>mend(Option)Insufficientevidence tore<strong>com</strong>mend(Option)Insufficientevidence tore<strong>com</strong>mend(Option)Insufficientevidence tore<strong>com</strong>mend(Option)Insufficientevidence tore<strong>com</strong>mend(Option)Insufficientevidence tore<strong>com</strong>mend(Option)Circadian phasemarkersInsufficientevidence tore<strong>com</strong>mend(Option)Insufficientevidence tore<strong>com</strong>mend(Option)Insufficientevidence tore<strong>com</strong>mend(Option)Insufficientevidence tore<strong>com</strong>mend(Option)Indicated(Option)Insufficientevidence tore<strong>com</strong>mend(Option)Actigraphy fordiagnosisIndicated(Option)Not routinelyindicated(Option)Indicated(Guideline)Indicated(Guideline)Indicated(Option)Indicated(Option)Actigraphy forresponse totherapyIndicated(Guideline)Indicated(Guideline)Indicated(Guideline)Indicated(Guideline)Indicated(Guideline)Indicated(Guideline)Sleep log ordiaryIndicated(Guideline)Indicated(Guideline)Indicated(Guideline)Indicated(Guideline)Indicated(Guideline)Indicated(Guideline)TherapyPlanned SleepSchedulesIndicated(Standard)Indicated(Option)Indicated(Option)Indicated(Option)Indicated(Option)Mixed modalityindicated (Option/Guideline)*Timed LightExposureIndicated(Guideline)Indicated(Option)Indicated(Option)Indicated(Guideline)Indicated(Option)Indicated(Option)Timed MelatoninAdministrationIndicated(Guideline)Indicated(Standard)Indicated(Option)Indicated(Guideline)Indicated Sighted(Option)Indicated Unsighted(Guideline)Indicatedfor certainpopulation #(Option)HypnoticsIndicated(Guideline)Indicated(Option)- Not Re<strong>com</strong>mended(Option)- -StimulantsIndicated Caff(Option)Indicated(Option)- - - -Alerting Agents 9Indicated(Guideline)- - - - -* Mixed modality therapy may be effective in elderly-demented/Nursing Home ISWR patients (Guideline) or those with moderate to severe mentalretardation (Option)#Timed melatonin may be effective in those with moderate to severe mental retardation, but is not re<strong>com</strong>mended at present for elderly-demented/Nursing Home patients (Option)Caff = caffeine; Sighted=sighted persons; Unsighted=unsighted persons; - = no re<strong>com</strong>mendation formulated due to lack of evidence.SLEEP, Vol. 30, No. 11, 2007 1448Practice Parameters for the Clinical Evaluation of CRSD—Morgenthaler et al
- Page 1 and 2:
Practice Management Tips ForSHIFT W
- Page 3 and 4:
Patient QuestionnaireDo you often f
- Page 5 and 6:
Sleep/Wake LogIn bedOut of bedLight
- Page 7 and 8:
PHQ-9 QUICK DEPRESSION ASSESSMENTFo
- Page 9 and 10:
Insomnia Severity IndexPlease answe
- Page 11 and 12:
Take-Away PointsSHIFT WORK DISORDER
- Page 13 and 14:
SHIFT WORKDISORDERBright Light Ther
- Page 40 and 41:
PrimarycareScreeningfor depressioni
- Page 42 and 43:
PrimarycareThescreening questionnai
- Page 44 and 45:
Shift-work disorderContents and Fac
- Page 46 and 47:
Shift-work disorderThe diagnosis of
- Page 48 and 49:
Shift-work disorderas heightened le
- Page 50 and 51:
Shift-work disorderFigure 1 Risk ra
- Page 52 and 53:
Shift-work disorderare not function
- Page 54 and 55:
The characterization andpathology o
- Page 56 and 57:
Shift-work disorderFigure 2 Sleep/w
- Page 58 and 59:
Shift-work disorderFigure 3 Blood p
- Page 60 and 61:
Recognition of shift-workdisorder i
- Page 62 and 63:
Shift-work disorderThe timing of sh
- Page 64 and 65:
Shift-work disorderthe other potent
- Page 66 and 67:
Managing the patient withshift-work
- Page 68 and 69:
Shift-work disorderFigure 3 Optimal
- Page 70 and 71:
Shift-work disorderfor a motor vehi
- Page 72 and 73: Shift-work disordermoderate caffein
- Page 74 and 75: Supplement toAvailable at jfponline
- Page 76 and 77: Armodafinil for Treatment of Excess
- Page 78 and 79: Armodafinil for Treatment of Excess
- Page 80 and 81: Armodafinil for Treatment of Excess
- Page 82 and 83: Armodafinil for Treatment of Excess
- Page 84 and 85: Armodafinil for Treatment of Excess
- Page 86 and 87: Armodafinil for Treatment of Excess
- Page 88 and 89: Armodafinil for Treatment of Excess
- Page 90 and 91: Armodafinil for Treatment of Excess
- Page 92 and 93: Armodafinil for Treatment of Excess
- Page 94 and 95: Armodafinil for Treatment of Excess
- Page 96 and 97: Armodafinil for Treatment of Excess
- Page 98 and 99: The Epidemiology and Diagnosis of I
- Page 100 and 101: The Epidemiology and Diagnosis of I
- Page 102 and 103: The Epidemiology and Diagnosis of I
- Page 120 and 121: CIRCADIAN RHYTHM SLEEP DISORDERSPra
- Page 124 and 125: 3.2.1.1 Both the Morningness-Evenin
- Page 126 and 127: Five studies used one of the newer
- Page 128 and 129: as an indicator of phase in sighted
- Page 130 and 131: 4.4 Advanced Sleep Phase DisorderBe
- Page 132 and 133: 45. Walsh, JK, Randazzo, AC, Stone,
- Page 134: 123. Van Someren, EJ, Kessler, A, M
- Page 142 and 143: Table 1—Subject Demographicsn M:F
- Page 144 and 145: Scale. 28 The simple reaction time
- Page 146 and 147: Median RT (msec)1600A14001200100080
- Page 148 and 149: 10Mentally AExhaustedSharpScore8642
- Page 150 and 151: Current Treatment Options in Neurol
- Page 152 and 153: 398 Sleep Disordersand sleep loss,
- Page 154 and 155: 400 Sleep DisordersTable 1. Treatme
- Page 156 and 157: 402 Sleep DisordersStandard dosageC
- Page 158 and 159: 404 Sleep DisordersStandard procedu
- Page 160 and 161: 406 Sleep DisordersCaffeineMelatoni
- Page 162 and 163: 408 Sleep DisordersWake-promoting a
- Page 164 and 165: 410 Sleep Disordersnight shift: ada