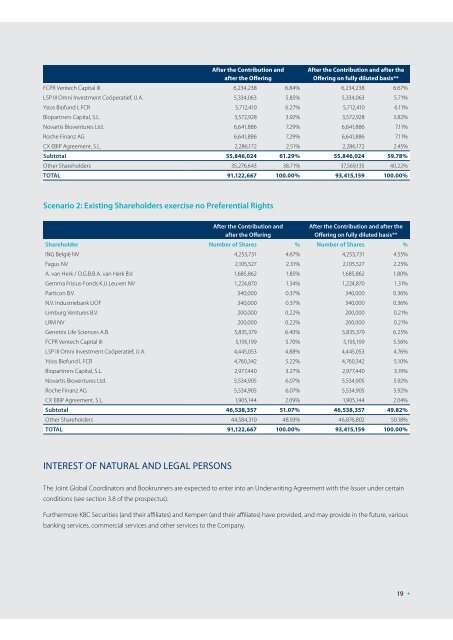
After the Contribution andafter the OfferingAfter the Contribution and after theOffering on fully diluted basis**FCPR Ventech Capital III 6,234,238 6.84% 6,234,238 6.67%LSP III Omni Investment Coöperatief, U.A. 5,334,063 5.85% 5,334,063 5.71%Ysios Biofund I, FCR 5,712,410 6.27% 5,712,410 6.11%Biopartners Capital, S.L. 3,572,928 3.92% 3,572,928 3.82%Novartis Bioventures Ltd. 6,641,886 7.29% 6,641,886 7.11%Roche Finanz AG 6,641,886 7.29% 6,641,886 7.11%CX EBIP Agreement, S.L. 2,286,172 2.51% 2,286,172 2.45%Subtotal 55,846,024 61.29% 55,846,024 59.78%Other Shareholders 35,276,643 38.71% 37,569,135 40.22%TOTAL 91,122,667 100.00% 93,415,159 100.00%Scenario 2: Existing Shareholders exercise no Preferential RightsAfter the Contribution andafter the OfferingAfter the Contribution and after theOffering on fully diluted basis**Shareholder Number of Shares % Number of Shares %ING België NV 4,253,731 4.67% 4,253,731 4.55%Fagus NV 2,105,527 2.31% 2,105,527 2.25%A. van Herk / O.G.B.B.A. van Herk B.V. 1,685,862 1.85% 1,685,862 1.80%Gemma Frisius-Fonds K.U.Leuven NV 1,224,870 1.34% 1,224,870 1.31%Particon B.V. 340,000 0.37% 340,000 0.36%N.V. Industriebank LIOF 340,000 0.37% 340,000 0.36%Limburg Ventures B.V. 200,000 0.22% 200,000 0.21%LRM NV 200,000 0.22% 200,000 0.21%Genetrix Life Sciences A.B. 5,835,379 6.40% 5,835,379 6.25%FCPR Ventech Capital III 5,195,199 5.70% 5,195,199 5.56%LSP III Omni Investment Coöperatief, U.A. 4,445,053 4.88% 4,445,053 4.76%Ysios Biofund I, FCR 4,760,342 5.22% 4,760,342 5.10%Biopartners Capital, S.L. 2,977,440 3.27% 2,977,440 3.19%Novartis Bioventures Ltd. 5,534,905 6.07% 5,534,905 5.92%Roche Finanz AG 5,534,905 6.07% 5,534,905 5.92%CX EBIP Agreement, S.L. 1,905,144 2.09% 1,905,144 2.04%Subtotal 46,538,357 51.07% 46,538,357 49.82%Other Shareholders 44,584,310 48.93% 46,876,802 50.18%TOTAL 91,122,667 100.00% 93,415,159 100.00%Interest of Natural and Legal PersonsThe Joint Global Coordinators and Bookrunners are expected to enter into an Underwriting Agreement with the Issuer under certainconditions (see section 3.8 of the prospectus).Furthermore KBC Securities (and their affiliates) and Kempen (and their affiliates) have provided, and may provide in the future, variousbanking services, commercial services and other services to the Company.19 •
CalendarPublication in the Belgian Financial Press and in the Belgian State Gazette of thenotice required by Article 593 of the Companies Codeat the latestT-8at the latestMay 4, 2011Determination of the Issuance Price and Ratio T-1 May 11, 2011Separation of coupon no. 1 (representing the Preferential Right) after closing of the markets T May 12, 2011Availability to the public of the prospectus T May 12, 2011Listing of the Contribution Shares on the regulated market of Euronext Brussels T+1 May 13, 2011Trading of Shares ex-Right T+1 May 13, 2011Opening date of the subscription with Preferential Rights T+1 May 13, 2011Listing of the Preferential Rights on the regulated market of Euronext Brussels T+1 May 13, 2011Closing date of the subscription with Preferential Rights T+15 May 27, 2011End of listing of the Preferential Rights on the regulated market of Euronext Brussels T+15 May 27, 2011Announcement via press release of the results of the Rights Offering before opening of the markets T+18 May 30, 2011Accelerated private placement of the Scrips T+19 May 31, 2011Pricing and allocation of the Scrips T+19 May 31, 2011Announcement via press release of the results of the Scrips Private Placement T+19 May 31, 2011Publication in the Belgian Financial Press of the results of the Offering and of the amount due to holdersT+20 June 1, 2011of unexercised Preferential RightsPayment of the Issuance Price by or on behalf of the subscribers T+25 June 6, 2011Realisation of the capital increase T+25 June 6, 2011Delivery of the New Shares to the subscribers T+25 June 6, 2011Listing of the New Shares on the regulated market of Euronext Brussels T+25 June 6, 2011Payment to holders of unexercised Preferential Rights as of T+28 June 9, 2011Definition of the main terms used in this summary and elsewherein the prospectusThroughout this summary and elsewhere in the prospectus, certain terms and expressions are used. Unless the context in which theseterms and expressions are used does not so permit, or unless these terms or expressions are defined differently, they should be readand understood as follows:Articles of AssociationBelgian Financial PressBoard of DirectorsCellerixCellerix Shareholders InvestmentCellerix Shareholders’ Lock-up UndertakingsCETClosing date of the OfferingClosing date of the Rights OfferingCompanies CodeThe articles of association of <strong>TiGenix</strong>.De Tijd.The board of directors of <strong>TiGenix</strong>.Cellerix S.A., a corporation incorporated under the laws of Spain, having itsregistered office at Calle Marconi, 1, Parque Tecnológico de Madrid, Tres Cantos,28760 Madrid, Spain, registered with the Commercial Registry of Madrid undervolume number 20117, page 81, sheet M-355159 and with tax identification number(C.I.F.) A-84008986.The equity investment in Cellerix collectively made by certain Cellerix investorsbetween April 26, 2011 and the Contribution Date through a cash contribution toCellerix in the amount of €18,155,669.74 in exchange for 3,431,425 Cellerix shares.The lock-up undertakings by the holders of Contribution Shares included in theContribution Agreement and in the Lock-up Agreement.Central European Time.The day on which the capital increase pursuant to the Offering is realised. This date isexpected to be June 6, 2011.Last day on which the Existing Shareholders and the other investors with PreferentialRights can subscribe to the New Shares. This date is expected to be May 27, 2011.The Belgian Companies Code of May 7, 1999, as amended.20 • <strong>TiGenix</strong> • Rights Offering
- Page 2 and 3: TiGenix NV(Public limited liability
- Page 4 and 5: Table of ContentsSummary ..........
- Page 7 and 8: 3.7.1 Categories of potential inves
- Page 9 and 10: 5.7.1 Shares and warrants held by i
- Page 11 and 12: 7.5.2 Taxation.....................
- Page 13 and 14: SummaryThe words written in capital
- Page 15 and 16: Activities and strategy of the Comp
- Page 17 and 18: • Two allogeneic adult stem cell
- Page 19 and 20: • TiGenix’ success depends on i
- Page 22 and 23: Unaudited pro forma income statemen
- Page 24 and 25: Recent developmentsAcquisition of C
- Page 26 and 27: In this context, we would like to s
- Page 28 and 29: Announcement of the results of theO
- Page 32 and 33: ContributionContribution AgreementC
- Page 34 and 35: Risk factorsAny investment in the P
- Page 36 and 37: commercialisation of ChondroCelect,
- Page 38 and 39: in delays in bringing products to t
- Page 40 and 41: The Company cannot predict what eff
- Page 42 and 43: y each of its patents and patent ap
- Page 44: to exercise preferential subscripti
- Page 47 and 48: this prospectus which is capable of
- Page 49 and 50: • investment professionals fallin
- Page 51 and 52: 1. General information and informat
- Page 53 and 54: 1.4.2 Company documents and otherin
- Page 55 and 56: • the granting of discharge of li
- Page 57 and 58: • in case of registered Shares, t
- Page 59 and 60: cast at the meeting. If the amount
- Page 61 and 62: No one may cast a greater number of
- Page 63 and 64: If a Belgian resident individual ne
- Page 65 and 66: Belgium has concluded tax treaties
- Page 67 and 68: 3. Information on the Contributiona
- Page 69 and 70: In this context, we would like to s
- Page 71 and 72: TiGenix has a limited financial deb
- Page 73 and 74: Thousands of Euro (€) TiGenix Cel
- Page 75 and 76: 3.5.2 Issuance Price and RatioThe i
- Page 77 and 78: (c)Rules for subscriptionSubject to
- Page 79 and 80: The Scrips Private Placement will o
- Page 81 and 82:
to comply with any of its obligatio
- Page 83 and 84:
3.11.2 Scenario 1: Existing Shareho
- Page 85 and 86:
• a pledge of Locked Shares to a
- Page 87 and 88:
4. General information about THECOM
- Page 89 and 90:
DateINCORPORATIONFebruary 21,2000Tr
- Page 91 and 92:
Upon completion of the IPO of TiGen
- Page 93 and 94:
Issue dateMay 14,2004April 20,2005T
- Page 95 and 96:
4.7.2 Voting rightsAs further descr
- Page 97 and 98:
can be obtained free of charge at t
- Page 99 and 100:
5.2.4 Composition of the Board of D
- Page 101 and 102:
Amonis. Mr. Duron has been CEO of K
- Page 103 and 104:
5.3.4 Nomination and remunerationco
- Page 105 and 106:
• investor relations: nurturing c
- Page 107 and 108:
The remuneration of the members of
- Page 109 and 110:
5.7.2 Shares and warrants held by e
- Page 111 and 112:
Cellerix EBIP 2010An EBIP for senio
- Page 113 and 114:
The minutes must also contain a jus
- Page 115 and 116:
6. Activities of Tigenix andits sub
- Page 117 and 118:
• Clinical stage pipeline. TiGeni
- Page 119 and 120:
6.3.3 Acquisition of CellerixAs a r
- Page 121 and 122:
Full thickness articular cartilage
- Page 123 and 124:
Fig. 6.2: Autologous Chondrocyte Im
- Page 125 and 126:
Preclinical work in a meniscus repa
- Page 127 and 128:
The Company is also ensuring high q
- Page 129 and 130:
Recognising the importance of pre-l
- Page 131 and 132:
In Spain, since the passage of Orde
- Page 133 and 134:
Fig. 6.6: ChondroMimetic procedure
- Page 135 and 136:
6.5.3 Commercial strategyBuilding o
- Page 137 and 138:
6.7 Manufacturing & logisticsEffici
- Page 139 and 140:
The international application WO06/
- Page 141 and 142:
ChondroMimetic competitionThe main
- Page 143 and 144:
in Cellerix’ GMP facility in Madr
- Page 145 and 146:
6.14.3 History and development of C
- Page 147 and 148:
The table below gives an overview o
- Page 149 and 150:
Prevalence No. of cases (2010) Esti
- Page 151 and 152:
Fig. 6.9: Platform development stra
- Page 153 and 154:
In developing Cx601, Cellerix has b
- Page 155 and 156:
6.14.6 Manufacturing & logisticsCel
- Page 157 and 158:
population, methods for the isolati
- Page 159 and 160:
• Cimzia (certolizumab) - UCB: Al
- Page 161 and 162:
Spanish Ministry of Education and S
- Page 163 and 164:
7.2 Consolidated income statementTw
- Page 165 and 166:
7.3.1.2 Research and development ex
- Page 167 and 168:
7.3.2.3 Selling, general and admini
- Page 169 and 170:
7.4.1 Cash flows from operating act
- Page 171 and 172:
Cash & cash equivalents and intangi
- Page 173 and 174:
As of December 31, 2010, the Group
- Page 175 and 176:
8. Consolidated Financial informati
- Page 177 and 178:
8.1.3 Consolidated cash flow statem
- Page 179 and 180:
8.1.5 Notes to consolidated financi
- Page 181 and 182:
operating results and operating pla
- Page 183 and 184:
The Group does not account for work
- Page 186 and 187:
8.1.5.4 Operating result (EBIT)Resu
- Page 188 and 189:
8.1.5.7 TaxesThere is no current ta
- Page 190 and 191:
8.1.5.10 Tangible assetsThousands o
- Page 192 and 193:
8.1.5.14 Deferred charges & accrued
- Page 194 and 195:
8.1.5.18 Finance lease obligations
- Page 196 and 197:
Business combination Cellerix SADes
- Page 198 and 199:
Weighted averageexercise priceTOTAL
- Page 200 and 201:
the respective issue date of the wa
- Page 202 and 203:
Transactions with non-executive dir
- Page 204 and 205:
8.1.5.28 Subsequent eventsAcquisiti
- Page 206 and 207:
8.1.5.31 Disclosure under Article 1
- Page 208 and 209:
Additional statementsThe preparatio
- Page 210 and 211:
9.1.2 Stand-alone balance sheetYear
- Page 212 and 213:
9.1.4 Stand-alone statement of chan
- Page 214 and 215:
) Standards and interpretations iss
- Page 216 and 217:
The clinical development of new dru
- Page 218 and 219:
future against which they may be ap
- Page 220 and 221:
Cellerix’ research and developmen
- Page 222 and 223:
YearThousandsof Euro2010 2009 2008E
- Page 224 and 225:
Thousands of Euro (€)Laboratoryeq
- Page 226 and 227:
Current financial assetsShown below
- Page 228 and 229:
At December 31, Cellerix’ share c
- Page 230 and 231:
9.1.5.17 Deferred revenueThe balanc
- Page 232 and 233:
9.1.5.21 Share-based paymentsThe EB
- Page 234 and 235:
GrantsCellerix received several gra
- Page 236 and 237:
10. Report regarding unaudited prof
- Page 238 and 239:
EMAFDAFibrous tissueGCPGMPGrowth fa
- Page 240 and 241:
Appendix 1: Press releases 2006-201
- Page 242 and 243:
Appendix 2: REGULATORY APPROVALPROC
- Page 244 and 245:
like ChondroCelect or the future ce
- Page 246 and 247:
TitleCountry/regionPatent/applicati
- Page 248 and 249:
TitleCountry/regionPatent/applicati
- Page 250 and 251:
Title“Cell populations having imu
- Page 252 and 253:
D. Trademarks of CellerixThe table
- Page 254 and 255:
Decision resources.Dell’Accio, F.
- Page 256 and 257:
Noyes, F. R., Barber-Westin, S. D.,
- Page 258 and 259:
Appendix 5: 2008, 2009 and2010 mana
- Page 260 and 261:
Contents1. The year in brief ......
- Page 262 and 263:
2. Financial informationa. The Inco
- Page 264 and 265:
The Directors shall call a Sharehol
- Page 266 and 267:
• Exercise price changed to 5.291
- Page 268 and 269:
10 • TiGenix • Rights Offering
- Page 270 and 271:
Contents1. The year in brief ......
- Page 272 and 273:
3. Financial informationa. The Inco
- Page 274 and 275:
Exceptionally, for the following re
- Page 276 and 277:
(d)(e)Duration: The Options Plan wi
- Page 278 and 279:
Contents1. The year in brief ......
- Page 280 and 281:
3. Financial informationa. The Inco
- Page 282 and 283:
eceipt sent to the number or addres
- Page 284 and 285:
THE COMPANYTiGenix NVRomeinse straa