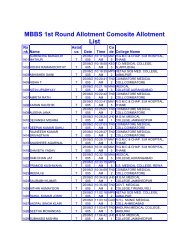
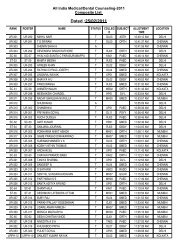
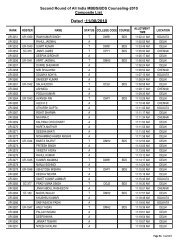
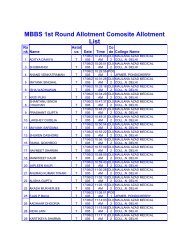
manual of methods of analysis of foods - Ministry of Health and ...
manual of methods of analysis of foods - Ministry of Health and ...
manual of methods of analysis of foods - Ministry of Health and ...
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
METALS<br />
(6) Repeat extraction by shaking cyclohexane layer with 3 ml <strong>of</strong> sodium<br />
hydroxide solution (h – ii) <strong>and</strong> add the aqueous layer into the content<br />
<strong>of</strong> the 50 ml beaker mentioned in step 5.<br />
(7) Wash the cyclohexane layer retained in the separatory funnel with 5<br />
ml <strong>of</strong> NaOAC solution (k) by cautiously tilting the funnel <strong>and</strong> forcing<br />
the contents to flow forth <strong>and</strong> back six times.<br />
(8) Decolourise the iodine present in the acidified aqueous solution (step<br />
6) by dropwise addition <strong>of</strong> ascorbic acid solution (i). Add 2 ml <strong>of</strong><br />
catechol violet solution (j) <strong>and</strong> mix. Add to this solution, the sodium<br />
acetate washing solution (obtained in step 7) <strong>and</strong> adjust the pH <strong>of</strong><br />
solution to 3.8 ± 0.1 with ammonia solution (1) <strong>and</strong>/or HCl solution<br />
(e), using pH meter.<br />
(9) Quantitatively transfer the solution (after adjusting the pH to 3.8 ± 0.1<br />
into a 25 ml volumetric flask, add 2 ml <strong>of</strong> absolute ethanol, make up<br />
to the mark with water <strong>and</strong> mix. Set the flask aside for 45 min at room<br />
temperature. Measure the absorbance ‘A’ <strong>of</strong> the solution using water<br />
as reference.<br />
Carry out a blank determination starting with 50 ml <strong>of</strong> 4.5M sulphuric acid<br />
solution (f) <strong>and</strong> carrying out steps from 2 to 9, the optical density <strong>of</strong> the<br />
blank should not exceed 0.070.<br />
Convert the absorbance ‘A’ to µg <strong>of</strong> Tin from calibration graph <strong>and</strong><br />
calculate Tin content <strong>of</strong> sample as follows:<br />
64