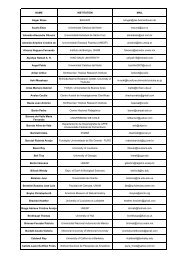
MARTIN THIEL ET AL.PERUIquique−20°Ant<strong>of</strong>agasta−25°−30°Coquimbo−30°Sou<strong>the</strong>rn latitude−33°CHILE−35°Concepción−40°1 2 3 4 5 6 7 8 9 10111213 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 29 30Figure 17 Break points <strong>of</strong> distribution along <strong>the</strong> north-<strong>central</strong> Chilean coast documented in 29 biogeographicalclassifications published between 1951 <strong>and</strong> 2006 (breaks outside <strong>the</strong> range 17–38°S are not shown). Thefigure includes a wide spectrum <strong>of</strong> animal <strong>and</strong> macroalgal taxa <strong>of</strong> varying hierarchical levels, life forms <strong>and</strong>habitats, <strong>and</strong> some classifications may partially overlap in one or more <strong>of</strong> <strong>the</strong>se categories. Each classificationis represented along an imaginary vertical axis in correspondence with <strong>the</strong> numbers at <strong>the</strong> bottom, wheredashed lines denote <strong>the</strong> zones in which break points were found (each one marked by a thick horizontal dash),ranging from one to three break points per study. Horizontal lines highlight <strong>the</strong> latitudes 30°S <strong>and</strong> 33°S wherebreak points tend to concentrate. Classifications (for numbers 1–10 <strong>and</strong> 11–22, see references in compilationsby Brattström & Johanssen 1983 <strong>and</strong> Camus 2001, respectively): 1, Mollusca (Carcelles & Williamson 1951);2, Foraminifera (Boltovskoy 1964); 3, Mollusca (Marincovich 1973); 4, several animal taxa (Knox 1960);5, eulittoral organisms (Hartmann-Schröder & Hartmann 1962); 6, intertidal Mollusca (Dell 1971); 7, benthicanimals (Semenov 1977); 8, several animal taxa (Viviani 1979); 9, Anthozoa (Sebens & Paine 1979); 10,several animal taxa (Brattström & Johanssen 1983); 11, benthic macroalgae (Santelices 1980); 12, Bryozoa(Moyano 1991); 13, fishes (Mann 1954); 14, Isopoda (Menzies 1962); 15, several animal taxa (Ekman 1953);16, several animal taxa (Balech 1954); 17, planktonic Euphausiids (Antezana 1981); 18, Asteroidea (Madsen1956); 19, macroalgae (Alveal et al. 1973); 20, s<strong>and</strong>y beach Isopoda (Jaramillo 1982); 21, Porifera Demospongiae(Desqueyroux & Moyano 1987); 22, several invertebrate taxa (Lancellotti & Vásquez 1999); 23,demersal fishes (Sielfeld & Vargas 1996); 24, littoral Teleostei (Ojeda et al. 2000); 25, Phaeophyta (Meneses& Santelices 2000); 26, Rhodophyta (Meneses & Santelices 2000); 27, several animal <strong>and</strong> algal taxa (Camus2001); 28, Peracarid crustaceans (Thiel 2002); 29, benthic Polychaeta (Hernández et al. 2005); 30, pelagicbarnacles (Hinojosa et al. 2006).variations <strong>of</strong> mollusc diversity along <strong>the</strong> HCS, which would be determined by shelf area (Valdovinoset al. 2003), while <strong>the</strong> distribution <strong>of</strong> some littoral species appears more related to regional variationsin temperature trends (Rivadeneira & Fernández 2005). Thus, <strong>the</strong> transitions in nor<strong>the</strong>rn Chile are256
THE HUMBOLDT CURRENT SYSTEM OF NORTHERN AND CENTRAL CHILEnot readily explained simply by <strong>the</strong> contact between warm <strong>and</strong> cold biotas, <strong>and</strong> proper explanationswill require a multivariate, integrative approach <strong>and</strong> an exploration <strong>of</strong> possible external forcings.The role <strong>of</strong> past <strong>and</strong> present processes in nor<strong>the</strong>rn-<strong>central</strong> Chile (18–35°S)Different lines <strong>of</strong> historical <strong>and</strong> ecological evidence suggest that nor<strong>the</strong>rn-<strong>central</strong> Chile constitutesa very complex <strong>and</strong> dynamical biogeographic scenario. Clearly, present-day patterns are notdivorced from physical changes related to <strong>the</strong> origin <strong>and</strong> installation <strong>of</strong> <strong>the</strong> cold HCS during <strong>the</strong>Tertiary or from subsequent Quaternary fluctuations (e.g., see Villagrán 1995, Hinojosa & Villagrán1997, Villa-Martínez & Villagrán 1997, Maldonado & Villagrán 2002). The establishment <strong>of</strong> <strong>the</strong>HCS had involved both <strong>the</strong> northward advance <strong>of</strong> <strong>the</strong> subantarctic biota <strong>and</strong> <strong>the</strong> northward retraction<strong>of</strong> a former tropical/subtropical biota (Brattström & Johanssen 1983, Camus 2001), with consequencesthat may persist until <strong>the</strong> present, reflected in <strong>the</strong> heterogeneous character <strong>of</strong> <strong>the</strong> nor<strong>the</strong>rnbiota. For instance, 10 <strong>of</strong> <strong>the</strong> nowadays most common bivalves in nor<strong>the</strong>rn Chile exhibit upper<strong>the</strong>rmal tolerances exceeding <strong>the</strong> highest temperatures recorded during EN events in <strong>the</strong> past century(Urban 1994), which is unexpected for species evolving in a cold upwelling <strong>system</strong>. At least one<strong>of</strong> <strong>the</strong>m (Argopecten purpuratus) is thought to be a relict <strong>of</strong> <strong>the</strong> Miocene tropical/subtropical fauna(Wolff 1987). Such physiological ‘anomalies’ suggest <strong>the</strong> presence <strong>of</strong> an inertial faunistic componentwithin <strong>the</strong> modern nor<strong>the</strong>rn biota (i.e., remnants <strong>of</strong> <strong>the</strong> former warm fauna that escaped <strong>the</strong> forcedretraction to lower latitudes, <strong>and</strong> maintained <strong>the</strong>ir warm-water characteristics facilitated by re<strong>current</strong>post-Miocene warming events such as EN) (Wolff 1987). In comparison, <strong>the</strong> marine flora appearsmore homogeneous <strong>and</strong> dominated mainly by subantarctic species, while tropical/subtropical speciesare virtually absent (Santelices & Meneses 2000). For instance, some common <strong>and</strong> ecologicallyimportant kelp species are not only more sensitive to warming episodes but also <strong>the</strong>ir upper <strong>the</strong>rmaltolerance varies in accordance with <strong>the</strong> <strong>the</strong>rmal latitudinal gradient (Martínez 1999).In this regard, <strong>the</strong> nor<strong>the</strong>rn fauna underwent repeated distributional alterations in <strong>the</strong> pastassociated with climatic fluctuations. Some <strong>of</strong> <strong>the</strong>m involved <strong>the</strong> simultaneous range retraction <strong>and</strong>expansion <strong>of</strong> different species (e.g., Ortlieb et al. 1994), but more <strong>of</strong>ten <strong>the</strong> occurrence <strong>of</strong> tropical/subtropical fauna related to warming (EN-like) events in <strong>the</strong> Pleistocene (e.g., Ortlieb 1995) <strong>and</strong>Holocene (e.g., Llagostera 1979). Moreover, Neogene processes related to <strong>the</strong> establishment <strong>of</strong> <strong>the</strong>modern upwelling <strong>system</strong> in <strong>the</strong> HCS (e.g., shallowing <strong>of</strong> <strong>the</strong> OMZ) provoked a mass extinction<strong>of</strong> bivalve molluscs (>75% <strong>of</strong> species), with lasting impacts on <strong>the</strong>ir <strong>current</strong> distribution patterns<strong>and</strong> biological characteristics (Rivadeneira 2005), <strong>and</strong> similar effects have been suggested for <strong>the</strong>polychaete fauna (R.A. Moreno et al. 2006a). Notwithst<strong>and</strong>ing, <strong>the</strong> causal relationships betweenhistorical events <strong>and</strong> <strong>current</strong> distribution patterns in nor<strong>the</strong>rn Chilean waters remain largely unexplored,although <strong>the</strong>ir importance may be overwhelming.On <strong>the</strong> o<strong>the</strong>r h<strong>and</strong>, modern processes also have strong influences in nor<strong>the</strong>rn Chile, particularlyinterannual fluctuations related to ENSO, which, however, should be looked at retrospectively,acknowledging <strong>the</strong> frequency <strong>and</strong> importance <strong>of</strong> EN-like events throughout <strong>the</strong> Holocene (e.g, seeMaldonado & Villagrán 2002). Strong EN events can modify <strong>the</strong> taxonomic composition <strong>of</strong> littoralcommunities (e.g., Arntz 1986, Castilla & Camus 1992, Camus et al. 1994, Vega et al. 2005,Vásquez et al. 2006) <strong>and</strong> <strong>the</strong> geographical occurrence <strong>of</strong> many species including key structuralcomponents such as intertidal <strong>and</strong> subtidal kelps (e.g., Lessonia nigrescens, L. trabeculata <strong>and</strong>Macrocystis pyrifera). Short-term modifications <strong>of</strong> community composition during EN occurthrough local extinctions <strong>and</strong> invasions, depending also on local conditions, which ei<strong>the</strong>r favour orprevent <strong>the</strong>ir occurrence (Arntz 1986, Camus et al. 1994, Martínez et al. 2003, Castilla et al. 2005a).Moreover, <strong>the</strong> impacts on key species may scale up to produce long-term biogeographic changes,as exemplified by <strong>the</strong> dramatic effects <strong>of</strong> <strong>the</strong> 1982–1983 EN event on <strong>the</strong> intertidal kelp Lessonia257