PDF file - Facultatea de Chimie şi Inginerie Chimică
PDF file - Facultatea de Chimie şi Inginerie Chimică
PDF file - Facultatea de Chimie şi Inginerie Chimică
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
MATHEMATICAL MODELING FOR THE CRYSTALLIZATION PROCESS OF HYDROXYAPATITE …<br />
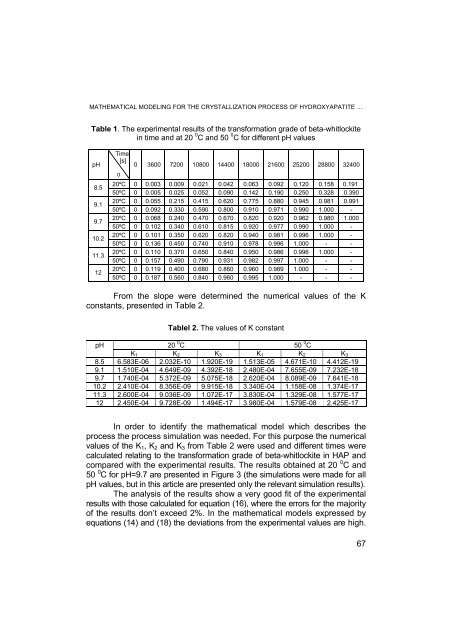
Table 1. The experimental results of the transformation gra<strong>de</strong> of beta-whitlockite<br />
in time and at 20 0 C and 50 0 C for different pH values<br />
pH<br />
Time<br />
[s]<br />
η<br />
0 3600 7200 10800 14400 18000 21600 25200 28800 32400<br />
8.5<br />
20ºC<br />
50ºC<br />
0<br />
0<br />
0.003<br />
0.005<br />
0.009<br />
0.025<br />
0.021<br />
0.052<br />
0.042<br />
0.090<br />
0.063<br />
0.142<br />
0.092<br />
0.190<br />
0.120<br />
0.250<br />
0.158<br />
0.328<br />
0.191<br />
0.390<br />
9.1<br />
20ºC<br />
50ºC<br />
0<br />
0<br />
0.055<br />
0.092<br />
0.215<br />
0.330<br />
0.415<br />
0.590<br />
0.620<br />
0.800<br />
0.775<br />
0.910<br />
0.880<br />
0.971<br />
0.945<br />
0.990<br />
0.981<br />
1.000<br />
0.991<br />
-<br />
9.7<br />
20ºC<br />
50ºC<br />
0<br />
0<br />
0.068<br />
0.102<br />
0.240<br />
0.340<br />
0.470<br />
0.610<br />
0.670<br />
0.815<br />
0.820<br />
0.920<br />
0.920<br />
0.977<br />
0.962<br />
0.990<br />
0.980<br />
1.000<br />
1.000<br />
-<br />
10.2<br />
20ºC<br />
50ºC<br />
0<br />
0<br />
0.101<br />
0.136<br />
0.350<br />
0.450<br />
0.620<br />
0.740<br />
0.820<br />
0.910<br />
0.940<br />
0.978<br />
0.981<br />
0.996<br />
0.996<br />
1.000<br />
1.000<br />
-<br />
-<br />
-<br />
11.3<br />
20ºC<br />
50ºC<br />
0<br />
0<br />
0.110<br />
0.157<br />
0.370<br />
0.490<br />
0.650<br />
0.790<br />
0.840<br />
0.931<br />
0.950<br />
0.982<br />
0.986<br />
0.997<br />
0.996<br />
1.000<br />
1.000<br />
-<br />
-<br />
-<br />
12<br />
20ºC<br />
50ºC<br />
0<br />
0<br />
0.119<br />
0.187<br />
0.400<br />
0.560<br />
0.680<br />
0.840<br />
0.860<br />
0.960<br />
0.960<br />
0.995<br />
0.989<br />
1.000<br />
1.000<br />
-<br />
-<br />
-<br />
-<br />
-<br />
From the slope were <strong>de</strong>termined the numerical values of the K<br />
constants, presented in Table 2.<br />
Tablel 2. The values of K constant<br />
20 0 C 50 0 pH<br />
C<br />
K1 K2 K3 K1 K2 K3<br />
8.5 6.583E-06 2.032E-10 1.920E-19 1.513E-05 4.671E-10 4.412E-19<br />
9.1 1.510E-04 4.649E-09 4.392E-18 2.480E-04 7.655E-09 7.232E-18<br />
9.7 1.740E-04 5.372E-09 5.075E-18 2.620E-04 8.089E-09 7.641E-18<br />
10.2 2.410E-04 8.356E-09 9.915E-18 3.340E-04 1.158E-08 1.374E-17<br />
11.3 2.600E-04 9.036E-09 1.072E-17 3.830E-04 1.329E-08 1.577E-17<br />
12 2.450E-04 9.728E-09 1.494E-17 3.980E-04 1.579E-08 2.425E-17<br />
In or<strong>de</strong>r to i<strong>de</strong>ntify the mathematical mo<strong>de</strong>l which <strong>de</strong>scribes the<br />
process the process simulation was nee<strong>de</strong>d. For this purpose the numerical<br />
values of the K1, K2 and K3 from Table 2 were used and different times were<br />
calculated relating to the transformation gra<strong>de</strong> of beta-whitlockite in HAP and<br />
compared with the experimental results. The results obtained at 20 0 C and<br />
50 0 C for pH=9.7 are presented in Figure 3 (the simulations were ma<strong>de</strong> for all<br />
pH values, but in this article are presented only the relevant simulation results).<br />
The analysis of the results show a very good fit of the experimental<br />
results with those calculated for equation (16), where the errors for the majority<br />
of the results don’t exceed 2%. In the mathematical mo<strong>de</strong>ls expressed by<br />
equations (14) and (18) the <strong>de</strong>viations from the experimental values are high.<br />
67