Prevention Guide - Safe Handling of Hazardous Drugs - Irsst
Prevention Guide - Safe Handling of Hazardous Drugs - Irsst
Prevention Guide - Safe Handling of Hazardous Drugs - Irsst
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
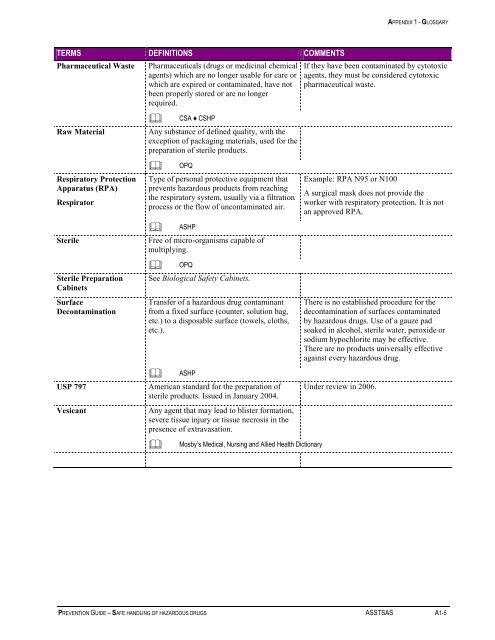
APPENDIX 1 - GLOSSARY<br />
TERMS DEFINITIONS COMMENTS<br />
Pharmaceutical Waste<br />
Raw Material<br />
Respiratory Protection<br />
Apparatus (RPA)<br />
Respirator<br />
Sterile<br />
Sterile Preparation<br />
Cabinets<br />
Surface<br />
Decontamination<br />
USP 797<br />
Vesicant<br />
Pharmaceuticals (drugs or medicinal chemical<br />
agents) which are no longer usable for care or<br />
which are expired or contaminated, have not<br />
been properly stored or are no longer<br />
required.<br />
<br />
CSA ♦ CSHP<br />
Any substance <strong>of</strong> defined quality, with the<br />
exception <strong>of</strong> packaging materials, used for the<br />
preparation <strong>of</strong> sterile products.<br />
<br />
OPQ<br />
Type <strong>of</strong> personal protective equipment that<br />
prevents hazardous products from reaching<br />
the respiratory system, usually via a filtration<br />
process or the flow <strong>of</strong> uncontaminated air.<br />
<br />
ASHP<br />
Free <strong>of</strong> micro-organisms capable <strong>of</strong><br />
multiplying.<br />
<br />
OPQ<br />
See Biological <strong>Safe</strong>ty Cabinets.<br />
Transfer <strong>of</strong> a hazardous drug contaminant<br />
from a fixed surface (counter, solution bag,<br />
etc.) to a disposable surface (towels, cloths,<br />
etc.).<br />
<br />
ASHP<br />
American standard for the preparation <strong>of</strong><br />
sterile products. Issued in January 2004.<br />
Any agent that may lead to blister formation,<br />
severe tissue injury or tissue necrosis in the<br />
presence <strong>of</strong> extravasation.<br />
<br />
Mosby’s Medical, Nursing and Allied Health Dictionary<br />
If they have been contaminated by cytotoxic<br />
agents, they must be considered cytotoxic<br />
pharmaceutical waste.<br />
Example: RPA N95 or N100<br />
A surgical mask does not provide the<br />
worker with respiratory protection. It is not<br />
an approved RPA.<br />
There is no established procedure for the<br />
decontamination <strong>of</strong> surfaces contaminated<br />
by hazardous drugs. Use <strong>of</strong> a gauze pad<br />
soaked in alcohol, sterile water, peroxide or<br />
sodium hypochlorite may be effective.<br />
There are no products universally effective<br />
against every hazardous drug.<br />
Under review in 2006.<br />
PREVENTION GUIDE – SAFE HANDLING OF HAZARDOUS DRUGS ASSTSAS A1-5