Prevention Guide - Safe Handling of Hazardous Drugs - Irsst
Prevention Guide - Safe Handling of Hazardous Drugs - Irsst
Prevention Guide - Safe Handling of Hazardous Drugs - Irsst
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
2. RISKS RELATED TO THE USE OF HAZARDOUS DRUGS<br />
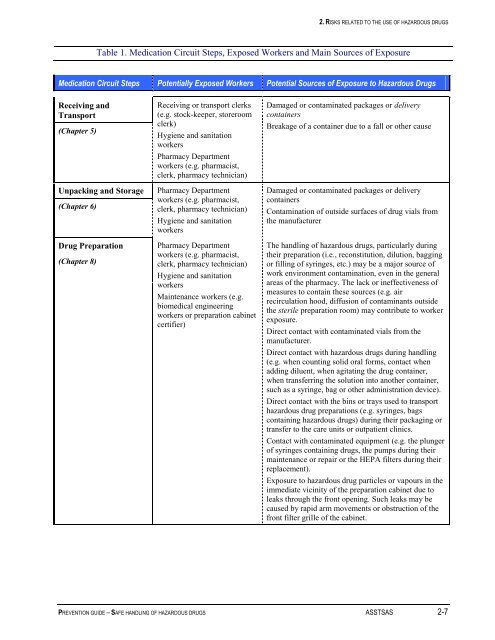
Table 1. Medication Circuit Steps, Exposed Workers and Main Sources <strong>of</strong> Exposure<br />
Medication Circuit Steps Potentially Exposed Workers Potential Sources <strong>of</strong> Exposure to <strong>Hazardous</strong> <strong>Drugs</strong><br />
Receiving and<br />
Transport<br />
(Chapter 5)<br />
Unpacking and Storage<br />
(Chapter 6)<br />
Drug Preparation<br />
(Chapter 8)<br />
Receiving or transport clerks<br />
(e.g. stock-keeper, storeroom<br />
clerk)<br />
Hygiene and sanitation<br />
workers<br />
Pharmacy Department<br />
workers (e.g. pharmacist,<br />
clerk, pharmacy technician)<br />
Pharmacy Department<br />
workers (e.g. pharmacist,<br />
clerk, pharmacy technician)<br />
Hygiene and sanitation<br />
workers<br />
Pharmacy Department<br />
workers (e.g. pharmacist,<br />
clerk, pharmacy technician)<br />
Hygiene and sanitation<br />
workers<br />
Maintenance workers (e.g.<br />
biomedical engineering<br />
workers or preparation cabinet<br />
certifier)<br />
Damaged or contaminated packages or delivery<br />
containers<br />
Breakage <strong>of</strong> a container due to a fall or other cause<br />
Damaged or contaminated packages or delivery<br />
containers<br />
Contamination <strong>of</strong> outside surfaces <strong>of</strong> drug vials from<br />
the manufacturer<br />
The handling <strong>of</strong> hazardous drugs, particularly during<br />
their preparation (i.e., reconstitution, dilution, bagging<br />
or filling <strong>of</strong> syringes, etc.) may be a major source <strong>of</strong><br />
work environment contamination, even in the general<br />
areas <strong>of</strong> the pharmacy. The lack or ineffectiveness <strong>of</strong><br />
measures to contain these sources (e.g. air<br />
recirculation hood, diffusion <strong>of</strong> contaminants outside<br />
the sterile preparation room) may contribute to worker<br />
exposure.<br />
Direct contact with contaminated vials from the<br />
manufacturer.<br />
Direct contact with hazardous drugs during handling<br />
(e.g. when counting solid oral forms, contact when<br />
adding diluent, when agitating the drug container,<br />
when transferring the solution into another container,<br />
such as a syringe, bag or other administration device).<br />
Direct contact with the bins or trays used to transport<br />
hazardous drug preparations (e.g. syringes, bags<br />
containing hazardous drugs) during their packaging or<br />
transfer to the care units or outpatient clinics.<br />
Contact with contaminated equipment (e.g. the plunger<br />
<strong>of</strong> syringes containing drugs, the pumps during their<br />
maintenance or repair or the HEPA filters during their<br />
replacement).<br />
Exposure to hazardous drug particles or vapours in the<br />
immediate vicinity <strong>of</strong> the preparation cabinet due to<br />
leaks through the front opening. Such leaks may be<br />
caused by rapid arm movements or obstruction <strong>of</strong> the<br />
front filter grille <strong>of</strong> the cabinet.<br />
PREVENTION GUIDE – SAFE HANDLING OF HAZARDOUS DRUGS ASSTSAS 2-7