CMDITR Review of Undergraduate Research - Pluto - University of ...
CMDITR Review of Undergraduate Research - Pluto - University of ...
CMDITR Review of Undergraduate Research - Pluto - University of ...
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
Photodegradation Conditions and Mechanism <strong>of</strong> the “EZ”- FTC<br />
Chromophore<br />
Olga Kondratets, Shoreline Community College<br />
Yi Liao and Bruce Robinson<br />
Robinson Lab, Dept. <strong>of</strong> Chemistry, <strong>University</strong> <strong>of</strong> Washington<br />
Objective/Thesis<br />
Recently, research <strong>of</strong> the organic molecules<br />
that exhibit electro optic (EO) non-linear<br />
properties has gained a lot <strong>of</strong> attention. The<br />
reason for this interest is their possible use in the<br />
construction <strong>of</strong> new materials and devices that<br />
utilize organic molecules and promise to be fast,<br />
effective, and inexpensive relative to traditional<br />
inorganic materials. 1<br />
Currently, chromophores are the most<br />
promising organic molecules, exhibiting large<br />
EO non-linear properties both on small and large<br />
scales. For this research we chose to study the<br />
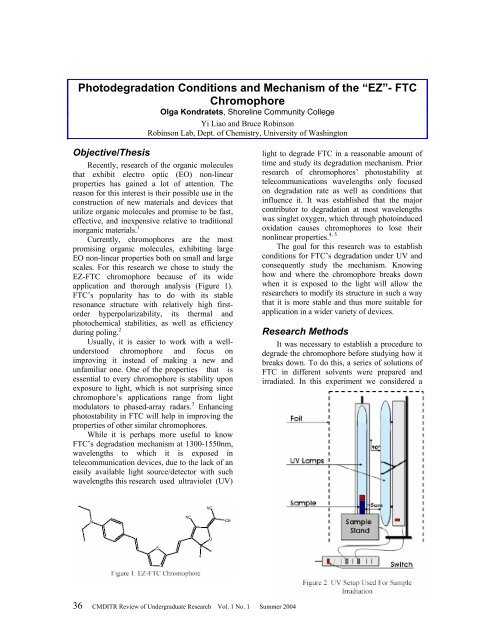
EZ-FTC chromophore because <strong>of</strong> its wide<br />
application and thorough analysis (Figure 1).<br />
FTC’s popularity has to do with its stable<br />
resonance structure with relatively high firstorder<br />
hyperpolarizability, its thermal and<br />
photochemical stabilities, as well as efficiency<br />
during poling. 2<br />
Usually, it is easier to work with a wellunderstood<br />
chromophore and focus on<br />
improving it instead <strong>of</strong> making a new and<br />
unfamiliar one. One <strong>of</strong> the properties that is<br />
essential to every chromophore is stability upon<br />
exposure to light, which is not surprising since<br />
chromophore’s applications range from light<br />
modulators to phased-array radars. 3 Enhancing<br />
photostability in FTC will help in improving the<br />
properties <strong>of</strong> other similar chromophores.<br />
While it is perhaps more useful to know<br />
FTC’s degradation mechanism at 1300-1550nm,<br />
wavelengths to which it is exposed in<br />
telecommunication devices, due to the lack <strong>of</strong> an<br />
easily available light source/detector with such<br />
wavelengths this research used ultraviolet (UV)<br />
light to degrade FTC in a reasonable amount <strong>of</strong><br />
time and study its degradation mechanism. Prior<br />
research <strong>of</strong> chromophores’ photostability at<br />
telecommunications wavelengths only focused<br />
on degradation rate as well as conditions that<br />
influence it. It was established that the major<br />
contributor to degradation at most wavelengths<br />
was singlet oxygen, which through photoinduced<br />
oxidation causes chromophores to lose their<br />
nonlinear properties. 4, 5<br />
The goal for this research was to establish<br />
conditions for FTC’s degradation under UV and<br />
consequently study the mechanism. Knowing<br />
how and where the chromophore breaks down<br />
when it is exposed to the light will allow the<br />
researchers to modify its structure in such a way<br />
that it is more stable and thus more suitable for<br />
application in a wider variety <strong>of</strong> devices.<br />
<strong>Research</strong> Methods<br />
It was necessary to establish a procedure to<br />
degrade the chromophore before studying how it<br />
breaks down. To do this, a series <strong>of</strong> solutions <strong>of</strong><br />
FTC in different solvents were prepared and<br />
irradiated. In this experiment we considered a<br />
36 <strong>CMDITR</strong> <strong>Review</strong> <strong>of</strong> <strong>Undergraduate</strong> <strong>Research</strong> Vol. 1 No. 1 Summer 2004