MRI indicates that bone marrow stem cells remain grafted in <strong>the</strong> infarcted rat heart for sixteen weeks,but do not improve cardiac functionStuckey DJ 1 , Carr CA 1 , Martin-Rendon E 2 , Tyler DJ 1 , Willmott C 2 , Cassidy PJ 1 , Hale SJM 2 , Schneider JE 1 , Tatton L 2 ,Harding SE 3 , Radda GK 1 , Watt S 2 , Clarke K 11 Cardiac Metabolism Research Group, Laboratory <strong>of</strong> Physiology, Anatomy and Genetics, <strong>University</strong> <strong>of</strong> Oxford.2 Stem Cell Research Laboratory, National Blood Service, John Radcliffe Hospital. Oxford.3 National Heart and Lung Institute, Imperial College School <strong>of</strong> Science, Technology and Medicine, London.O21IntroductionA promising, novel approach to <strong>the</strong> treatment <strong>of</strong> myocardial infarction(MI) and prevention <strong>of</strong> heart failure is cell grafting in <strong>the</strong> damagedmyocardium 1 To optimise stem cell <strong>the</strong>rapy a non-invasive methodthat can show <strong>the</strong> tissue distribution <strong>of</strong> <strong>the</strong> administered cells andmonitor cardiac function is required 2 . We have used iron-labelling <strong>of</strong>bone marrow stromal cells (BMSCs) to non-invasively track celllocation in <strong>the</strong> infarcted rat heart over 16 weeks using cine-MRI andto isolate <strong>the</strong> donor cells from <strong>the</strong> grafted hearts using <strong>the</strong> magneticproperties <strong>of</strong> <strong>the</strong> injected BMSCs. Cardiac function was measuredfrom 1 to 16 weeks post MI and BMSC administration.MethodsBMSCs were isolated from rat bone marrow, characterised by flowcytometry, transduced with lentiviral vectors expressing GFP andlabelled with 0.9 µm iron particles (Bangs Laboratories Inc). Ratswere anaes<strong>the</strong>tized and MI was induced by ligation <strong>of</strong> <strong>the</strong> left anteriordescending coronary artery. Ten minutes post MI, ei<strong>the</strong>r a total <strong>of</strong> 5 ×10 5 BMSCs (n = 8), or saline (n = 7), was injected into 4 separatelocations in <strong>the</strong> infarct periphery. Cardiac cine-MRI was performed at1, 4, 10 and 16 weeks post MI using an 11.7 T MR system with aBruker console and a 60 mm birdcage coil as described previously 3 .A stack <strong>of</strong> contiguous 1.5 mm true short axis ECG and respiratorygated cine images (FOV, 51.2 mm 2 ; matrix size 256×256; TE/TR,1.43/4.6 ms; 17.5° pulse; 25 to 35 frames per cardiac cycle) wereacquired to cover <strong>the</strong> entire left ventricle. Cell distribution andcardiac function were measured in each slice. At 16 weeks, heartswere removed, fixed and imaged ex vivo using high resolution 3D MRmicroscopy (fast gradient echo, FOV, 20 mm 3 ; matrix size,256×256×512; TR/TE, 1.8/30 ms), <strong>the</strong>n sectioned for histology ordigested to single cells using collagenase for donor cell re-isolation.ResultsSignal voids in <strong>the</strong> cardiac MR images caused by <strong>the</strong> iron particles in<strong>the</strong> BMSCs were detected in all rats at all times (Fig 1A). MRmicroscopy identified hypointense regions at <strong>the</strong> same position asthose identified in vivo (Fig 1B). In mildly infarcted hearts, <strong>the</strong>volume <strong>of</strong> <strong>the</strong> signal void decreased over <strong>the</strong> 16 weeks (Fig 1C),whilst <strong>the</strong> signal void volume did not decrease significantly inseverely infarcted hearts. When non-labelled BMSCs or naked Bangsiron particles were injected into <strong>the</strong> heart, no signal voids wereidentified after <strong>the</strong> first week. Donor cells containing iron particlesand expressing GFP were identified in MR-targeted heart sections andafter magnetic cell separation from digested hearts. Approximately1.5% <strong>of</strong> administered BMSCs were identified at 16 weeks. Themajority <strong>of</strong> donor cells maintained <strong>the</strong>ir BMSC phenotype, but on rareoccasions, fluorescently labelled cells with a cardiomyocytephenotype were identified.No improvements in left ventricular ejection fraction (Fig 2), strokevolume, cardiac output or reduction in non-contractile scar regionwere found between <strong>the</strong> control and BMSC treated animals at any <strong>of</strong><strong>the</strong> time points studies.Ejection Fraction (%)80706050400 5 10 15WeeksMI +BMSCsn = 8MIn = 7shamn = 6Figure 2: Left ventricular ejection fraction from rats that underwentei<strong>the</strong>r MI, MI + BMSC injection, or sham operation.ConclusionMRI can be used to track cells labelled with iron particles in damagedtissue for at least 16 weeks after injection and to guide tissuesectioning by accurately identifying regions <strong>of</strong> cell engraftment. Themagnetic properties <strong>of</strong> <strong>the</strong> iron labelled donor cells can be used for<strong>the</strong>ir isolation from host tissue to enable future characterisation. Inthis study, although <strong>the</strong> grafted BMSCs remained present in <strong>the</strong>infarcted hearts for 16 weeks, <strong>the</strong>y were unable to improve cardiacfunction.References1 Ma<strong>the</strong>r & Martin, 2004, Lancet 364 p 183-922 Bartunek et al, 2006, Eur Heart J, 11, p1338-403Tyler et al, 2006, J Cardiovas Magn Reson, 8, 327-33This project was funded by <strong>the</strong> British Heart FoundationFigure 1: In vivo (A) and ex vivo (B) MR images <strong>of</strong> iron labelled BMSCs in <strong>the</strong> infarcted rat heart 16 weeks after administration. By tenweeks <strong>the</strong> signal void volume arising from <strong>the</strong> donor cells was significantly larger in <strong>the</strong> hearts with greater damage (C).
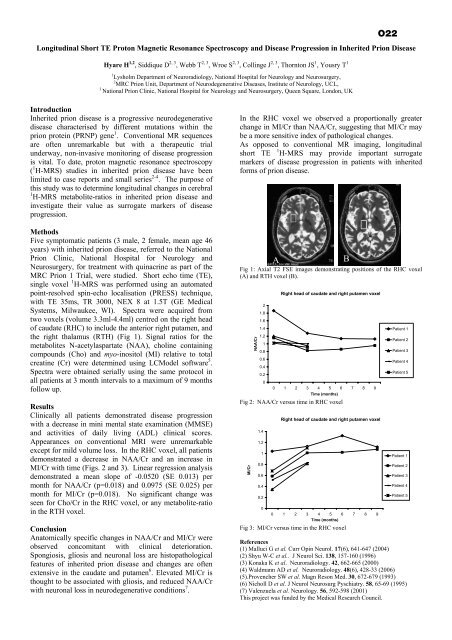
Longitudinal Short TE Proton Magnetic Resonance Spectroscopy and Disease Progression in Inherited Prion DiseaseHyare H 1,2 , Siddique D 2, 3 , Webb T 2, 3 , Wroe S 2, 3 , Collinge J 2, 3 , Thornton JS 1 , Yousry T 11 Lysholm <strong>Department</strong> <strong>of</strong> Neuroradiology, National Hospital for Neurology and Neurosurgery,2 MRC Prion Unit, <strong>Department</strong> <strong>of</strong> Neurodegenerative Diseases, Institute <strong>of</strong> Neurology, UCL,3National Prion Clinic, National Hospital for Neurology and Neurosurgery, Queen Square, London, UKO22IntroductionInherited prion disease is a progressive neurodegenerativedisease characterised by different mutations within <strong>the</strong>prion protein (PRNP) gene 1 . Conventional MR sequencesare <strong>of</strong>ten unremarkable but with a <strong>the</strong>rapeutic trialunderway, non-invasive monitoring <strong>of</strong> disease progressionis vital. To date, proton magnetic resonance spectroscopy( 1 H-MRS) studies in inherited prion disease have beenlimited to case reports and small series 2-4 . The purpose <strong>of</strong>this study was to determine longitudinal changes in cerebral1 H-MRS metabolite-ratios in inherited prion disease andinvestigate <strong>the</strong>ir value as surrogate markers <strong>of</strong> diseaseprogression.MethodsFive symptomatic patients (3 male, 2 female, mean age 46years) with inherited prion disease, referred to <strong>the</strong> NationalPrion Clinic, National Hospital for Neurology andNeurosurgery, for treatment with quinacrine as part <strong>of</strong> <strong>the</strong>MRC Prion 1 Trial, were studied. Short echo time (TE),single voxel 1 H-MRS was performed using an automatedpoint-resolved spin-echo localisation (PRESS) technique,with TE 35ms, TR 3000, NEX 8 at 1.5T (GE MedicalSystems, Milwaukee, WI). Spectra were acquired fromtwo voxels (volume 3.3ml-4.4ml) centred on <strong>the</strong> right head<strong>of</strong> caudate (RHC) to include <strong>the</strong> anterior right putamen, and<strong>the</strong> right thalamus (RTH) (Fig 1). Signal ratios for <strong>the</strong>metabolites N-acetylaspartate (NAA), choline containingcompounds (Cho) and myo-inositol (MI) relative to totalcreatine (Cr) were determined using LCModel s<strong>of</strong>tware 5 .Spectra were obtained serially using <strong>the</strong> same protocol inall patients at 3 month intervals to a maximum <strong>of</strong> 9 monthsfollow up.ResultsClinically all patients demonstrated disease progressionwith a decrease in mini mental state examination (MMSE)and activities <strong>of</strong> daily living (ADL) clinical scores.Appearances on conventional MRI were unremarkableexcept for mild volume loss. In <strong>the</strong> RHC voxel, all patientsdemonstrated a decrease in NAA/Cr and an increase inMI/Cr with time (Figs. 2 and 3). Linear regression analysisdemonstrated a mean slope <strong>of</strong> -0.0520 (SE 0.013) permonth for NAA/Cr (p=0.018) and 0.0975 (SE 0.025) permonth for MI/Cr (p=0.018). No significant change wasseen for Cho/Cr in <strong>the</strong> RHC voxel, or any metabolite-ratioin <strong>the</strong> RTH voxel.ConclusionAnatomically specific changes in NAA/Cr and MI/Cr wereobserved concomitant with clinical deterioration.Spongiosis, gliosis and neuronal loss are histopathologicalfeatures <strong>of</strong> inherited prion disease and changes are <strong>of</strong>tenextensive in <strong>the</strong> caudate and putamen 6 . Elevated MI/Cr isthought to be associated with gliosis, and reduced NAA/Crwith neuronal loss in neurodegenerative conditions 7 .In <strong>the</strong> RHC voxel we observed a proportionally greaterchange in MI/Cr than NAA/Cr, suggesting that MI/Cr maybe a more sensitive index <strong>of</strong> pathological changes.As opposed to conventional MR imaging, longitudinalshort TE1 H-MRS may provide important surrogatemarkers <strong>of</strong> disease progression in patients with inheritedforms <strong>of</strong> prion disease.Fig 1: Axial T2 FSE images demonstrating positions <strong>of</strong> <strong>the</strong> RHC voxel(A) and RTH voxel (B).NAA/Cr21.81.61.41.210.80.60.40.20Right head <strong>of</strong> caudate and right putamen voxel0 1 2 3 4 5 6 7 8 9Time (months)Fig 2: NAA/Cr versus time in RHC voxelMI/Cr1.41.210.80.60.40.20ARight head <strong>of</strong> caudate and right putamen voxel0 1 2 3 4 5 6 7 8 9Time (months)Fig 3: MI/Cr versus time in <strong>the</strong> RHC voxelPatient 1Patient 2Patient 3Patient 4Patient 5Patient 1Patient 2Patient 3Patient 4Patient 5References(1) Malluci G et al. Curr Opin Neurol. 17(6), 641-647 (2004)(2) Shyu W-C et al.. J Neurol Sci. 138, 157-160 (1996)(3) Konaka K et al. Neuroradiology. 42, 662-665 (2000)(4) Waldmann AD et al. Neuroradiology. 48(6), 428-33 (2006)(5).Provencher SW et al. Magn Reson Med. 30, 672-679 (1993)(6) Nicholl D et al. J Neurol Neurosurg Pyschiatry. 58, 65-69 (1995)(7) Valenzuela et al. Neurology. 56, 592-598 (2001)This project was funded by <strong>the</strong> Medical Research Council.B